 |
Renewable and Sustainable Energy Reviews
Bioethanol from macroalgae: Prospects and challenges |
 |
aEnergy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560012, India.
bCentre for Sustainable Technologies, Indian Institute of Science, Bangalore, 560012, India
*Corresponding author: Ramachandra T.V emram.ces@courses.iisc.ac.in
Deepthi Hebbale , deepthih@iisc.ac.in
|
|
1. Introduction
Fossil fuels such as oil, coal and natural gas are the major commercial energy sources and about 87% of global CO2 emitted due to the anthropogenic activities [1,2] are contributed by utilization of coal (43%), oil (36%) and natural gas (20%). Earth endows finite source of oil reserve and its increased consumption in several sectors has led to increased oil production, exerting pressure on the reserves which is apprehended to peak and no longer suffice the world’s demand with the fast dwindling stock [3]. Rising population with higher consumption levels coupled with a fast pace of development have spurred higher exploitation of fossil fuels leading to the escalating prices and resultant greenhouse gases (GHGs) posing problems for planet’s climatic stability [4–6]. It is imperative to ensure energy security through the sustainable alternative energy sources [7,8]. Globally, nations are actively addressing the issues concerning greenhouse gases and peak oil crisis through several mitigation measures such as; energy conservation, fuel substitution, incentives for the use of unconventional and renewable oil, and policy reforms such as carbon tax [8]. Therefore, the current focus is on carbon neutral renewable sources, notably photovoltaic, wind, hydrogen, etc. These alternative sources were useful in addressing the electricity requirement, but the exploration for viable alternatives to oil in order to meet the requirement of transport sector, etc. is quintes sential. Despite the existence of possible solutions such as renewable resources, energy efficient products (CFLs and LEDs) have not been widely adopted due to market barriers. Wind power contributes 2.5% of world electricity output and are weather dependent, susceptible to geographic and climatic changes [9,10]. Dependency on conventional generation coupled with the depleting stock and the enhanced envi ronmental awareness in the public have been the major constraints faced by the land based energy systems [11,12]. Nuclear power witnessed 2% growth in Europe, but encountered resistance with respect to disposal of waste, safety during nuclear accident and declining global uranium stocks. Nuclear disaster at Fukushima Daiichi nuclear power plant in 2011 [13], led Germany to rethink its energy policy [14].
India has been the 3rd largest energy consumer surpassing Russia, China and USA and about 80% of India’s energy consumption was contributed by imported crude oil [15], which was estimated to be 213.93 Million Metric Ton (MMT) in the year 2016–17. This is attributed to the poorly endowed natural reserve of hydrocarbon in India, however crude oil production in India is about 36.01 MMT, from the 0.3% oil reserves [15]. India is emerging as the fastest growing economy next to China with the growing energy demand, burgeoning population (at 1.58% annual) and dwindling stock of fossil fuel in next few decades, it is challenging to support this growing economy demand [16]. The total CO2 emission in India accounts for 965.9Tg/yr, with electricity generation (343Tg/yr) and transport (246.23Tg/yr) sectors as the major contributors [17]. The higher level of CO 2 emissions necessitates implementation of efficient management strategies to mitigate changes in climate [18]. The new renewable energy resources are being explored to meet the energy demand in all sectors and also research is underway to address the intermittency problems associated with wind and solar based energy systems [19–21].
In this context, studies have shown that biofuels are emerging as promising alternative to liquid fuels. Realizing the potential of biomass, different technologies have evolved towards the conversion of biomass into fuels, popularly known as biofuels [22–24]. Produced from renewable plant sources or other organic wastes, biofuels have the ad vantages of cutting down carbon emission and dependency on oil [25]. In India, around 80% of rural energy [26] is met by biomass energy consumption, in the form of firewood, agriculture residues, cow dung cake and other natural feedstock [24,27,28]. Fig. 1 represents the share of each country in the global bioethanol production, which highlights that India’s share is only 2% [29] despite burgeoning demand for fossil fuel. This emphasizes the need for augmentation with the viable indig enous alternative feedstock to minimize fossil fuel dependence.
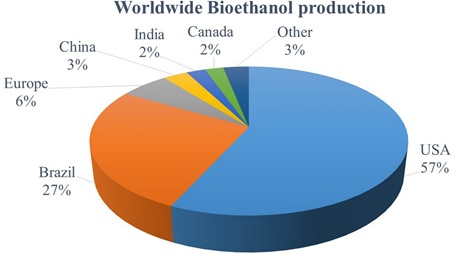 Fig. 1. Worldwide bioethanol production.
Fig. 1. Worldwide bioethanol production.
Biofuel from first generation feedstock involved food crops like corn and sugarcane which were exploited for biofuel production over three decades, but this technique encountered resistance due to the limited stock and competition with food crops [25]. The inadequacy of first generation feedstock in augmenting the growing energy demand led to the evolution of second generation feedstock involving lignocellulose biomass (Fig. 2). However, biofuel from second-generation feedstock also failed, due to the difficulty in scaling up and process technology involved in the cost-intensive delignification process [30]. Due to this, the cost of production of cellulosic ethanol is two to three fold higher than the price of corn grain ethanol [31]. In the US, it was seen that, the fossil energy required to produce bioethanol from corn, grain, soybean and wood biomass was more than the energy content of the biofuel, while sufficing only 12% of gasoline and 6% of the diesel demand. Though first and second generation feedstock are explored for biofuel production and assessed for carbon sequestration, environmental im pacts and production potential only marginally complies with various other sustainability criteria’s such as; disruption of global food supply, soil erosion, extensive usage of fertilizers, conversion of ecologically vulnerable wetlands, rainforests, peat lands, savannas into energy crop lands contributing to several magnitude of CO 2[32,33]. GHGs footprint of major cities in India [34], recorded aggregation of carbon dioxide equivalent emission of GHGs in the range of 13,734.59-38,633.2 Gg, with transportation being one of the major sector next to the energy generation. Emergence of a strong global biofuel feedstock is expected to realize a positive balance between energy and ecological footprints [35].
Table 1 illustrates the prospects of algal biomass emerging as an ideal alternative to the first and second generation [37,38]. Though, algae is being utilized as an energy feedstock since 1950s [26], the oil crisis of 1970’s spurted the research [39]. Algal feedstock do not require prime agricultural land and can be grown in fresh water, wastewater [40] and saline waters with zero nutrient input and non-interference with the land used for food production [38,41]. Algal biomass have higher photosynthetic efficiency (up to 5%) as compared to terrestrial biomass (1.8–2.2%) [42], and require for their growth light, carbon dioxide and nutrients (such as nitrogen, phosphorus, potassium, etc.), which are maintained through continuous flow of water [42]. Algae have a higher yield per unit area compared to terrestrial plants e.g. brown algae under the cultured condition, yields ~13.1 kg dry weight/m2 over 7 months as compared to sugarcane yield of ~10 kg dry weight/m2/yr [43]. Algae based on their morphology and size are grouped into micro and macroalgae [29]. Microalgae accumulate large quantities of neutral lipids which serves as raw material for biodiesel production [44,45], whereas macroalgae are carbohydrate rich biomass which are useful for bioethanol production. Large scale cultivation of macroalgae in Korea reveals an uptake of 8–10 tonne CO 2 per hectare [42].
| Biofuel |
Crop |
Yield(ton/ha/yr) |
Ethanol (litres/ha/yr) |
| First generation |
| Sugarcane |
50–90 |
3500–8000 |
| Sweet sorghum |
45–80 |
1750-5300 |
| Sugar beet |
15-50 |
1350-5500 |
| Fodder Beet |
100-200 |
4400-9350 |
| Wheat |
1.5-2.1 |
510-714 |
| Barley |
1.2-2.5 |
300-625 |
| Rice |
2.5-5.0 |
1075-2150 |
| Irish potatoes |
10-25 |
1110-2750 |
| Cassava |
10-65 |
1700-11050 |
| Sweet potatoes |
8-50 |
1336-8350 |
| Grapes |
10-25 |
1300-3250 |
| Second generation |
| Nipa palm |
|
2300-8000 |
| Maize |
1.7-5.4 |
600-1944 |
| Sorghum |
1.0-3.7 |
350-1295 |
| Third generation |
Algal biomass |
730 |
23400 |
Table 1 Yield and Ethanol production of First, Second and Third generation feedstocks.
1.1. Potential macroalgal feedstock available
Marine macroalgae or seaweeds establish on hard substratum and grow luxuriantly along nutrient rich coastal zone (Fig. 3). One of the richest seaweed resources in the world is in Nova Scotia/Gulf of St. Lawrence area[46]. Global seaweed distribution can be summarized as:
(i) Least flora <200 Spp in latitudes >60 in both hemispheres
(ii) Moderate flora of 600–700 spp. that occur throughout warm and cold tropical and temperate regions
(iii) Highest flora of 900–1100 spp. occur in four regions Southern Australia, Mediterranean, Japan and Philippines.
Seaweed resources and their uses are well established across regions in the world. Red seaweeds are mostly utilized for extraction of hydrocolloid valuing $585 million [47] and source of food (e.g. Salads) valuing $5 billion [48] with Asia as its prime market [43]. Cultivation of macroalgae is a promising option as seventy percent of the Earth surface is covered by water [39,42,49,50], therefore in order to satisfy these industrial demands, macroalgae are cultivated in large scale, mainly of the genus Laminaria, Undaria, Poryphyra, Eucheuma, Enteromorpha and Gracilaria representing 76% of total macroalgae aquaculture production [51].
In recent years, algal genera of Kappaphycus, Gelidium, Gracilaria, Sargassum, Laminaria and Ulva (Fig. 2), are the promising potential feedstock for biofuel production in addition to the value added products for phycocolloids extraction, human food, cosmetics, fertilizer and other chemicals [52,53]. These algal feedstock have been chosen considering the availability and assessment of resources around the globe, ease of cultivation and harvesting. However, there is still scope to assess other potential macroalgal species based on their availability, biochemical composition and prospects for cultivation.
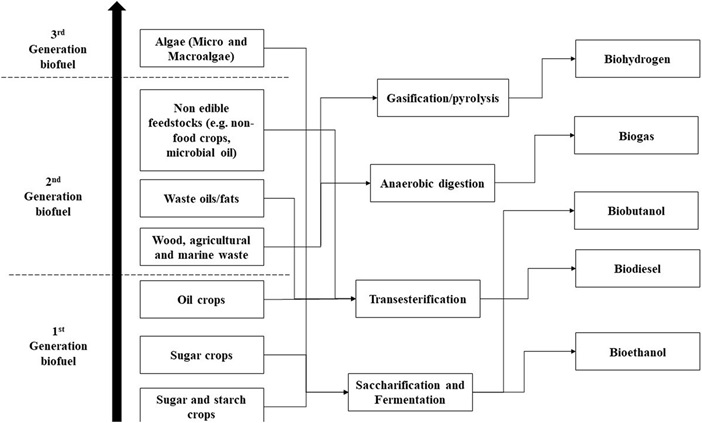 Fig. 2. Evolution of biofuel production from feedstocks and technologies.
Fig. 2. Evolution of biofuel production from feedstocks and technologies.
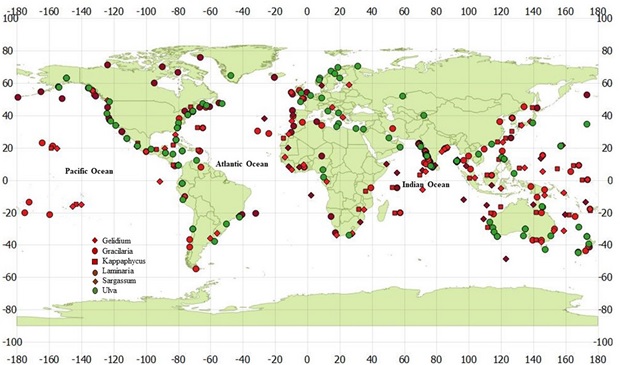 Fig. 3. Prominent coastal regions of the world rich in seaweed resources and potential feedstock for bioethanol production.
Fig. 3. Prominent coastal regions of the world rich in seaweed resources and potential feedstock for bioethanol production.
1.2. Bioethanol production from macroalgal feedstock
Bioethanol from algal biomass is a sustainable and eco-friendly option of renewable biofuel production [39]. Macroalgae or seaweed, saltwater thriving algae have proved to be the viable biofuel feedstock [54] for sustainable biofuel production as it avoids the competition with fresh water, food crops or cultivable land [39,55]. Seaweeds are multicellular marine macroalgae, broadly grouped as green, brown and red based on the pigment present in the thallus. Seaweed consists of carbohydrates (Table 2), which are converted to bioethanol by appropriate microorganisms such as yeast or bacteria. The common processes involved in ethanol production are
(i) pretreatment
(ii) hydrolysis
(iii) fermentation
| Characteristics |
Green seaweed |
Red seaweed |
Brown seaweed |
| Number of species recorded |
6032a |
7105b |
2039c |
| Habitat |
Freshwater and Marine |
Strictly marine |
Strictly marine |
| Photosynthetic pigment present |
Chlorophyll a, b, carotene and Xanthophyll |
Phycoerythrin |
Fucoxanthin |
| Photosynthetic rate (µmol CO2/h) g/dry |
30 to 1786 |
20–1808.7 |
100–500 |
| Productivity[dry g/(m2year)] |
7100 |
3300–11300 |
3300–11300 |
| Nature of cell wall |
Cellulose, pectin rarely hemi-cellulose |
Cellulose and pectic material with polysulphate esters |
Cellulose with alginic acid and fucocinic acid |
| Sexuality |
Isogamy to oogamy |
Advanced and complex (oogamous) |
Isogamy to oogamy |
| No. of flagella and their insertion |
2 or 4, equal anterior, whiplash |
Absent |
Only in reproductive cells, 2 unequal, lateral whiplash and tinsel |
| Cell structure Phycolibins |
Eukaryotic Absent |
Eukaryotic Allophycocyanin, r- Phycoerythrin r-Phycocyanin |
Eukaryotic Absent |
| Carotenoids |
α-, β-, γ- carotene |
α-, β- carotene |
α-, β- ε carotene |
| Xanthophylls |
Lutein Prasinoxanthin |
Lutein |
Fucoxanthin, Violaxanthin, Diadinoxanthin,Heteroxanthin, Vacheriaxanthin |
| Carbohydrate(%) |
30-60 |
30-50 |
20-30 |
| Protein (%) |
10-20 |
6-15 |
10-15 |
| Lipid (%) |
1-3 |
0.5-1.5 |
1-2 |
| Ash (%) |
13-22 |
5-15 |
14-28 |
| Photosynthetic reserve*(Stored food) |
Starch |
Floridean starch(intermediate between true starch and dextrin) |
Laminarin and mannitol (hexahydride alcohol) |
Source: [50,54,56–61].
Table 2 Detailed characteristics of different types of Seaweeds.
1.2.1. Pretreatment and hydrolysis for extraction of macroalgal sugar
Different types of biomass contain different amounts of sugars and the complexity of the biomass is reflected between structural and carbohydrate components [62,63]. Plant biomass is mostly composed of lignin (13.6–28.1%), cellulose (40.6–51.2%) and hemicellulose (28.5–37.2%) biopolymer [64], which serves as raw material for production of fuels. However, critical step involved in biofuel production is the conversion of biomass to sugars [65]. It is therefore important to carefully choose the pretreatment process based on the biomass and an optimal pretreatment process towards better yield of sugar with the low energy input [66].
Pretreatment involves physical, chemical and biological (or combinatorial) process to expose the cell constituents and cell wall materials of feedstock [67]. Physical pretreatment involves reduction in size of the feedstock to increase the surface area for better transport of acid/base catalysts, enzymes and steam to the fibers (cellulose) [68]. Chemical pretreatment involves dilute acid, alkaline, ammonia, organo solvent and other chemicals. Biological pretreatment involves microorganisms like bacteria and fungi (rich in cellulase enzyme) to degrade the biomass and release the sugars [69]. Integrated pretreatment involves combination of all the process such as acid catalyzed steam explosion, ammonium fiber explosion (AFEX), acid pretreated enzyme hydrolysis etc [66].
First generation biomass is starch based and requires no stringent pretreatment conditions to extract sugar, whereas lignocellulose biomass is complex in structure due to the presence of biopolymer lignin that embeds cellulose in a matrix resulting in a higher degree of polymerization and crystallization, which is the main factor responsible for recalcitrance [66,70–72] requiring a high cost for delignification process [73]. Therefore, the process of sugar extraction requires severe pretreatment conditions such as steam explosion at 200 C [74], at 121 C [75], AFEX, Sulphite pretreatment to overcome recalcitrance of lignocellulose (SPORL) [31], pressurized steam liquefaction [76]. It is seen that alkaline based pretreatment is effective in solubilizing significant portion of lignin from lignocellulose biomass [69]. Lignin was removed from cotton stalk pretreated using sodium hydroxide at high temperature and 96% fermentable sugars were recovered [77,78]. Around 11.4 MMT cotton plant wastes available in India, can generate 3533 billion litres of ethanol considering 90% fermentation efficiency [78]. Removal of 89% lignin and 69.77% hemicellulose in rice husk was achieved through wet air oxidation pretreatment method [79]. Hydrothermal pretreatment of wheat straw was carried out and viewed under scanning electron microscope (SEM), which reveal partial de-fibration of the lignin fibers due to pretreatment, whereas in delignification process lignin appears as layer of globular deposits exposing the cellulose structure [71].
Compared to this, macroalgae with the large concentration of structural polysaccharides (Table 3) and low lignin contents [80] requires mild and low-cost processes for extraction of sugars. The most widely used chemical pretreatment method for macroalgal biomass is dilute acid (Table 4), as it solubilizes hemicellulose and exposes cellulose fibers for further enzyme hydrolysis [68]. The energy consumed in acid pre-treatment is comparatively low as compared to other pre-treatments and higher sugar yields are achieved [69]. Dilute acid concentration for hydrolysis varies based on the feedstock, listed in Table 3. However, limitation of dilute acid pretreatment is the formation of Hydroxymethyl furfurals (HMF) and Levulinic acid (LA) resulting from the degradation of sugars that inhibit the subsequent process (fermentation) in ethanol production [81,82]. These inhibitors are mitigated by neutralization process before fermentation [83,84] or by employing other sustainable alternatives such as biological pretreatment: enzyme hydrolysis [53,85–94].
| |
Green seaweeds |
Red seaweeds |
Brown seaweeds |
| Structural polysaccharide |
Cellulose |
Cellulose, lignin |
Cellulose, Alginate |
| Storage polysaccharide |
Starch, Ulvan, Mannan |
Agar, Carrageenan |
Fucoidan, laminarin |
| Monosaccharides |
Glucose, Mannose, Rhamnose, Xylose, Galactose |
Glucose, Galactose, Agarose |
Glucose, Galactose, Fucose, Xylose |
| Sugar alcohol |
|
|
Mannitol |
| Sugar Acid |
Uronic acid, Glucuronic acid |
|
Uronic acid, Mannuronic acid,Glucuronic acid, Alginic acid |
Source [47–50]
Table 3 Sugar profile of Macroalgae.
| Green Seaweeds |
Pretreatment conditions |
Enzyme hydrolysis conditions |
Yeast/Bacterial strain and Fermentation process |
Reducing Sugar g/L |
Ethanol yield g/g |
Theoretical yield (%) |
Reference |
| E.intestinalis |
Hydrothermal process (75 mM for 90min) |
Celluclast 1.5 L and Viscozyme L (55 ° C, 120 rpm for 54 h) |
Saccharomyces cerevisiae KCTC 1126 (pH 5.5, 30 °C,220 rpm for 12 h) |
40.4 |
0.21 |
41.74 |
[120] |
| U.fasciata |
H2SO4 (0.1% at 100 °C for 1 h) |
Cellulase 22119 (Sodium acetate buffer pH 4.8 at 45 °C for 36 h) |
Saccharomyces cerevisiae(109 CFU/ml 28 °C,120 rpm for 48 h) |
20.6 |
0.45 |
88.24 |
[89] |
| U.lactuca |
|
Cellulase isolated from Cladosporium sphaerospermum (pH 4, 25 °C, 42 h) |
Saccharomyces cerevisiae MTCC180 (28 °C for 12 h) |
112 mg/g |
0.47 |
92.16 |
[94] |
| U.pertusa |
Citric acid buffer (0.1 M sterilized using autoclave) |
Meicelase (combined saccharification)(pH 5.5, 50 ° C, 100 rpm for 120 h) |
Saccharomyces cerevisiae IAM4178 (30 °C for 36 h) |
59.1 |
0.47 |
91.24 |
[70] |
| |
HTLP þ Enzyme (150 °C, 15 min) |
Cellulase&Amyloglucosidase(pH 4.8, 50 °C, 150 rpm for 24 h) |
Saccharomyces cerevisiae ATCC24858 (pH 5.5, 150 rpm, 30°C for 24 h) |
26 |
0.48 |
93.51 |
[97] |
| Red Seaweeds G. elegans |
|
Meicelase (pH 5.5 at 50°C for 120 h) |
Saccharomyces cerevisiae IAM4178 (30 °C for 36 h) |
49 |
0.38 |
73.63 |
[70] |
| G. amnasii |
H2SO4 (56–168mM,45–240min) |
Enzyme Viscozyme L (0.024 FBG/ml) |
Scheffersomyces stipitis (pH 5.5, 30 °C, 200 rpm) |
43.5 |
0.47 |
92.40 |
[108] |
| |
H2SO4 (2%, 150 °C for 4 h) |
|
Brettanomyces custersii KCCM11490 (pH 4.8–5.5, 27–30 °C) |
42.2 |
0.38 |
74.51 |
[126] |
| G. verrucosa |
H2SO4 (1.5%, at 80 °C for 2 h) |
|
|
87 |
0.43 |
84.29 |
[60] |
| |
373 mM H2SO4 |
Celluclast 1.5L and Viscozyme L(pH 5, 45 °C, 150 rpm for 72 h) |
Saccharomyces cerevisiae(30 °C for 48 h) |
20.4 |
0.48 |
94 |
[110] |
| Gracilaria sp. |
H2SO4 (0.1 N,121 °C for 30min) |
Commercial enzyme (pH 4.5,50 °C) |
Saccharomyces cerevisiae(30 °C for 48 h) |
11.46 |
0.42 |
82.80 |
[132] |
| K. alvarezii |
Soaked in 1.6 L distilled water for 30 min and boiled at 90 °C for 1 h |
Celluloclast 1.5 L & Novozyme(pH 5, 50 °C, 150 rpm for 24 h) |
Saccharomyces cerevisiae(pH 5,35 °C, 130 rpm for 6 h) |
79.2 |
0.25(SHF)
0.27(SSF) |
49
52.9 |
[88] |
| |
H2SO4 (0.2 M, 130 °C for 15min) |
|
Commercial brewer’s yeast (30 °C 120 rpm pH 5 for 72 h) |
20.4 |
0.21 |
41.18 |
[133] |
| |
H2SO4 (0.9 N, 100 °C for 1 h)5 cycles |
|
Saccharomyces cerevisiae NCIM (5% v/v, 30 °C 150 rpm, pH 6.4–6.8 for 48 h) |
51.9 |
0.42 |
82.36 |
[117] |
| P. palmata |
Acid hydrolysis |
|
|
21.84 |
0.173 |
33.92 |
[133] |
| Brown Seaweeds A. crassifolia |
Citric acid buffer (0.1 M sterilized using autoclave) |
Meicelase (5g/l at 50 °C for 120 h) |
Saccharomyces cerevisiae IAM 4178 (30 °C for 36 h) |
66.3 |
0.38 |
75 |
[70] |
| L. hyperborea |
Extracted in water at 65 °C |
|
Pichia angophorae |
30 |
0.43 |
84.31 |
[114] |
| |
Extracted in water 121 °C for 20min |
|
Zymobacter palmae (pH 6,30 °C) |
3.8 |
0.38 |
74.51 |
[115] |
| S. sagamianum |
|
|
Pichia stipitis (pH 5,200 rpm) |
19.8 |
0.35 |
69.32 |
[125] |
| S. janponica |
H2SO4 (1 mM, 121 °C, for 120min) |
Cellulase and cellobiase (pH 4.8, 50 °C, 150 rpm for 48 h) |
Saccharomyces cerevisiae (pH 6.5, 30 °C for 36 h) |
34 |
0.41 |
80.74 |
[103] |
| |
Acid hydrolysis (0.1 N, 121 °C for 15min) |
Celluclast 1.5L, Viscozyme L, Novoprime 959, Novoprime 969 or AMG 300L (50 °C, 150 rpm for 24 h) |
E.coli KO11 (30 °C for 24 h) |
30.54 |
0.41 |
80.39 |
[116] |
| |
H2SO4 (40 mM, 121 °C for 60min) |
Novozyme (Termamyl 120L) |
Pichia angophorae KCTC 17574 (5% 30 °C at 200 rpm, 136 h) |
45.6 |
0.16 |
33.3 |
[52] |
| |
Shredding and enzymatic (23 °C for 30min) |
|
Ethanol Red yeast (32 °C) |
35 |
0.45 |
88.24 |
[126] |
| U. pinnatifida |
Dilute acid (5% H2SO4, 120 °C for 24 h) |
Celluloclast 1.5L & Novozyme188 (pH 4.6, 45 °C) |
E.coli (pH 7170 rpm, 37 °;C for 12 h) |
20 |
0.144 |
28.2 |
[107] |
Table 4 Bioethanol production from macroalgal biomass.
Enzyme hydrolysis of cellulose is carried out efficiently by cellulolytic (cellulase) enzyme, which is comprised of exo-, endo-glucanases and cellobiase (β- D-glucosidase) enzymes [71]. Endoglucanases cleave cellulose at random sites of β-1, 4-bond and form free reducing ends and short-chain oligosaccharides [84] Exoglucanases cleaves the accessible ends of cellulose molecules to liberate glucose and cellobiose. β- D-glucosidase hydrolyses soluble cellobiose and other cellodextrin to produce glucose molecules [95]. Enzyme conversion is substrate specific without any by-product formation. The process could be enhanced [7], by exposing the cellulose fibres through pre-treatment using acid. Enzymatic hydrolysis disintegrates the cellulose and hemicellulose into simple sugars [96]. Along with this, depolymerization of xylan (polysaccharide composed of xylose) can be achieved by dilute-acid pretreatment [88] with about 64% xylose conversion efficiency. Pretreatment techniques include high thermal liquefaction process (HTLP) [97], alkali pretreatment, CaO, Ozonolysis, etc. However it was seen that the acid treated biomass was more susceptible for enzyme attack than HTLP, NaOH, CaO and other pretreatment [87].
Algal cell wall is composed of cellulose Iα (triclinic crystalline form) unlike the cellulose Iβ (monoclinic crystalline form) in plant cell wall. Cellulose Iα consists of weaker hydrogen bonds resulting from spatial arrangement of individual cellulose chains, resulting in easy access to endocellulases enzymes during enzyme hydrolysis [98]. Most common categories of enzymes considered for cell wall depolymerization are cellulases, hemicellulases and accessory enzymes [99], produced from wood-rot (soft rot) fungi such as Trichoderma, Penicillium, and Aspergillus [100]. The production costs of these enzymes are relatively higher. higher specific activity, achieved by genetic engineered strains [101]. Most common enzymes employed for seaweed hydrolysis are commercial enzymes such as Cellulase, Celluclast 1.5 L, Viscozyme L, Novozyme 188, Termamyl 120 L, β-glucosidase, Multifect, Meicelase, Amyloglucosidase etc operated at pH 4.5–5.5 and temperature 35–55 C, incubation time varies based on the algal feedstock [56,71,89, 90,102–109].
Cellulase producing microbes have been screened and isolated from various sources such as soil from forest and nature reserves, hot water springs, marine bacteria [90] compost, sewage, animal manure and bovine rumen [91]. Enzymatic hydrolysis has been done conventionally at >50 C, resulting in lower sugar yield [95]. Therefore, research is under progress for isolating efficient cellulolytic enzyme systems from a wide variety of bacteria, fungi, aerobes, anaerobes, mesophiles, thermophiles and thermo-stable microbes [92,93,96] which can overcome low sugar yield for biofuel production. Cellulase from thermophilic and psychrophilic microbes are preferred as they are resistant to high and low temperatures respectively [91]. Thermo-stable enzymes increase solubility of reactants and products, facilitating easy recovery of end products [96] while reducing hydrolysis time, decreasing contamination and cost of energy.
Marine fungus Cladosporium sphaerospermum was isolated to extract cellulase enzyme and used to hydrolyze U. pertusa biomass, which yielded 112 mg/g of reducing sugar at pH 4 and temperature 25 C for 42 h [94]. Similarly, marine bacteria was isolated from degrading U. lactuca to extract cellulase enzyme, which is tolerant to high salt concentration and alkaline pH [86]. Polysaccharolytic enzymes extracted from the gut of the abalone Haliotis midae degraded the polysaccharides laminarin, carboxymethylcellulose (CMC), alginate, agarose and carrageenan [109].
1.2.2. Fermentation of macroalgal sugars
Macroalgal biomass contain different types of polysaccharides, exclusively composed of glucose i.e., glucans. Main glucans present in green: cellulose and starch; red: cellulose and floridean starch; brown: cellulose and laminarin [46,50,61]. Non-glucans are sulphated polysaccharides such as agar, carrageenan and alginate. In order to obtain higher ethanol, hydrolysis of glucan as well as non-glucan with the fermentation of the resulting sugars is essential [60]. Sugar released from the pretreatment process has been fermented using microorganisms such as yeast, bacteria, and fungi, which ferment these sugars to produce ethanol as a by-product [41,110]. Saccharomyces cerevisiae is the commonly used yeast microorganism for fermentation as it readily ferments glucose [111]. However, pretreatment releases mixed sugars namely; glucose, galactose, mannitol, rhamnose and xylose. Due to the lack of xylose transport system, S.cerevisiae is not capable of utilizing xylose [112]. Its uptake takes place through glucose transport system and is regulated by the concentration of glucose. At only low concentration of glucose, xylose is consumed by the yeast [113]. As a result, studies related to isolation of wild yeast strains from various sources is done that can ferment both hexose and pentose sugars yielding higher ethanol. Bacteria, yeast and fungi are explored for xylose fermenting organisms, and mostly preferred organisms are bacteria and yeast as fungi are too slow for competitive industrial process [7].
Single or combination of strains are being attempted for utilization of sugars. Laminaran and mannitol obtained from L. hyperborea were subjected to fermentation using one bacterium (Zymobacter palmae T109) and three yeast strains (Pichia angophorae, Pacchysolen tannophilus and Kluyveromyces marxianus). It was seen that only P. angophorae is capable of fermenting laminaran and mannitol at higher oxygen transfer rate to produce 0.43 g ethanol/g substrate [114]. Utilization of mannitol by Zymobacter palmae resulted in the production of 0.37 g ethanol/g mannitol [115], however mannitol was utilized at lower oxygen rate in fermentation media. Mannitol was effectively fermented by E.coli KO11 for production of 0.41 g ethanol/g mannitol [116]. Similarly, glucuronic acid fermentation was attempted using Pachysolen tannophilus and E.coli.
Bioethanol production from all forms of macroalgal biomass; wet, dried and residues (after extraction of hydrocolloid) was attempted. Residues after extraction of hydrocolloids are rich in cellulose, which have been utilized for bioethanol production. Floating residue of L. japonica was subjected to acid pretreatment followed by enzyme hydrolysis, an ethanol yield of 14 g/L was obtained from 34 g/L of reducing sugar achieving 41.2% conversion efficiency [103]. Similarly, K. alvarezii dried residues after extraction of sap were utilized for production of bioethanol [117]. Wet biomass of G. amansii was used as bioethanol feedstock, Brettanomyces custersii KCTC 18154P strain was utilized for fermentation of the hydrolysate due to the ability of the strain in exhibiting co-fermentability. Utilization of raw or wet macroalgal biomass is not feasible for bioethanol production due to high viscosity of the medium for fermentation [118]. In green seaweeds, studies have focused on conversion of cellulose and starch to bioethanol. Whereas conversion of other sulphated polysaccharides such as Ulvan to produce ethanol is yet to be explored [60]. Non availability of natural strains capable of fermenting alginate, a major polysaccharide of brown algae [60], makes it difficult to achieve higher ethanol production.
Fermentation is carried out in two process, Separate Hydrolysis and Fermentation (SHF) and Simultaneous Saccharification and Fermentation (SSF) [119]. SHF involves hydrolysis and fermentation performed sequentially, whereas SSF involves performing simultaneous hydrolysis and fermentation [74]. Saccharina japonica, Undaria pinnatifida and Poryphyra were subjected to SSF using Pichia angophorae KCTC strain and obtained 7.7 g/L of ethanol [52]. SHF process is faster but presence of inhibitors resulting from acid pretreatment has significant impact on yeast microorganisms. SSF is preferred over SHF as the sugars released are readily metabolized by yeast microorganisms, which results in a faster ethanol production rate and lower capital costs. SSF has a drawback due to the difference in temperature optima of cellulase (50 C) and fermenting microorganism (35 C). SHF and SSF of Enteromorpha intestinalis or Ulva (Enteromorpha) intestinalis produced 8.6 g/L and 7.6 g/L with 30.5% and 29.6% fermentation efficiency respectively. Conversion of ethanol to acetic acid by yeast and suboptimal temperature of 30 C than the optimum temperature of 55 C for enzyme activity was attributed to the lower ethanol yield in SSF [120]. Higher temperature shortens the exponential phase of the yeast cell [121] affecting the ethanol production. However, this has been overcome through thermotolerant yeast strains or cell immobilization technique which allows higher processing temperatures [120–123]. Thermotolerant yeast species such as Candida tropicalis and Kluyveromyces marxianus (38–45 C) are mainly utilized to produce bioethanol from lignocellulosic biomass [123,124].
Bioethanol production from macroalgae utilized commercial yeast strains such as S. cerevisiae KCTC 1126 [110,111], MTCC 180 [60], IAM 4178 [70], ATCC 24858 [97], KCTC 17574 [52], Pichia stipitis [125], Pichia angophorae [114], Scheffersomyces stipitis [108], Brettanomyces custersii KCCM 11490 [126], Ethanol red yeast [36] and bacterial strains such as Zymobacter palmae [115] and Escherichia coli SJL2526 [107]. Fermentation of macroalgal polysaccharides is carried out at pH 4.5–6.8 and temperature 25–30 C and the incubation time is largely strain dependent. The yeast growth rate is dependent on temperature and fermentation time [131]. However, exponential phase of yeasts are shortened at large temperatures and pH > 4, requiring longer incubation for higher ethanol production, as reported in S.cerevisiae BY4742 [64]. Shorter fermentation time causes inadequate growth of microorganisms resulting in inefficient fermentation [132].
In order to optimize ethanol yield and improve substrate utilization range [129], studies focused on immobilization of yeast cells [121-130]. Immobilized yeast cells have enhanced the ethanol productivity and reusable for 15 cycles with bacterial cellulose-alginate sponge [121]. Free and immobilized strains were used for molasses fermentation. Free cells were unable to ferment at temperatures greater than 38 C, compared to immobilized yeast. Immobilized yeast strains exhibited both psychrophilic and thermo-tolerant characteristics, suitable for fermentation in a wide range of temperatures [131] and increased ethanol yield and higher cellular stability, while reducing downstream processing expenses [132]. Fermentation of U.lactuca biomass done using immobilized Saccharomyces cerevisiae strain, yielded ethanol (concentration of 12 g/g of sugar) with conversion efficiency of 47.1% [130]. Table 4 summarizes ethanol yield from the three types of macroalgae along with the process conditions and strains utilized for hydrolysis (pretreatment) and fermentation. Fermentation of red seaweed Gracilaria using free yeast cells yielded 0.41 g/g of ethanol and immobilized yeast cells yielded 0.42 g/g achieving 80 and 82.8% fermentation efficiency [132].
Studies emphasize on production of bioethanol from readily available carbohydrates of brown and red seaweeds, but utilization of red and brown seaweeds such as Kappaphycus, Gelidium, Gracilaria, Sargassum, and Laminaria have the likelihood to override the existing multi-billion dollar hydrocolloid industry [89]. This can be addressed in two ways: (i) utilization of cellulosic rich residue after hydrocolloid extraction, (ii) exploration of green seaweeds which are abundantly recorded from various estuaries and abandoned aquaculture ponds across maritime states in India [134]. Green seaweeds exhibit characteristics of a potential feedstock for biofuel production by their cosmopolitan distribution, wide environmental tolerance, higher growth rates and year around productivity [135]. In India, seaweeds are seldom consumed as a food source, and the suitability for biofuel production is still underexplored as several species accumulate different levels of carbohydrate. Seaweeds contain low amounts of polysaccharides composed of glucose, highlighting the need for ethanol production from carbohydrates including sulphated polysaccharides, sugar acids and sugar alcohols. Not all the reported microorganisms are capable of fermenting these sugars and a major limitation is lack of tractable microorganisms that can efficiently ferment all sugars extracted from seaweed into ethanol. Isolation of yeast strains to ferment both pentose (C5) and hexose (C6) sugars are vital for achieving high ethanol yield. In this backdrop, the current study explores bioethanol prospects from viable feedstock habituated in the west coast of India, which involves:
(i) screening and prioritizing potential macroalgal feedstock for bioethanol production based on the biochemical composition
(ii) comparative performance analysis of chemical and biological pretreatment method for extraction of sugar from macroalgal biomass; and
(iii) bioethanol potential assessment of green seaweeds and comparative analysis of ethanol yield across macroalgal species.
|
Citation :T.V. Ramachandra, Deepthi Hebbale, 2019, Bioethanol from macroalgae: Prospects and challenges, Available online 17 October 2019 1364-0321/© 2019 Elsevier Ltd. All rights reserved. https://doi.org/10.1016/j.rser.2019.109479 Received 24 February 2019; Received in revised form 23 September 2019; Accepted 8 October 2019
|




