Results and discussion
3.1. Role of microphyte (algae and bacteria) and macrophyte abundance and distribution in the nutrient dynamics
The physico-chemical alterations occurring spatio-temporally in lakes influence the incidence and activities of algal-bacterial systems in aquatic ecosystems, which has been observed in the present study. Urban lakes with the sustained inflow of untreated wastewater behaves as wastewater lagoons, with an initial anaerobic phase followed by an aerobic process (algal-bacterial symbiosis) where, the macromolecular complex biomolecules as polysaccharides, protein and lipids are first degraded by bacteria (Roche, 1998; Carta-Escobar et al., 2004). Subsequently, the algal systems are involved in key processes of i) reducing carbon (Mahapatra et al., 2013a) ii) inorganic nutrient (N and P) uptake (Mahapatra et al., 2013b) ii) storage of lipids (Ramachandra et al., 2009; Mahapatra and Ramachandra, 2013), proteins (Mahapatra et al., 2016) and carbohydrates (Ramachandra and Mahapatra, 2015) iv) pathogen removal by creating a high pH environment through algal photosynthesis (Mahapatra, 2015) and v) maintain the aerobic environment for the functioning of the treatment processes (Hosetti and Frost, 1998; Kirkwood et al., 2003; Chanakya et al., 2012, 2013; Mahapatra et al., 2013b). Higher species diversity, seasonal variations and species succession were noticed as these characteristics are a function of organic load, retention time, photoperiod, light intensities and predation by zooplanktons (Tharavathy and Hosetti, 2003; Ahmadi et al., 2005). The treatment levels and status of such kind of wastewater fed system have been assessed through investigations of the type of algae, their densities in terms of abundance; biomass and community structure (Sukias et al., 2001).
3.2. Seasonal variability's in nutrients and other parameters
At each sampling locations (Fig. 1), multiple samples were collected. Physico-chemical parameters were estimated for each sample and Tables 2 and 3 list season-wise water quality parameters. Monsoon corresponds to June to November months of Southwest and North-east monsoon period. pH values ranged from 7.4 8.2 in Bellandur Lake and the highest pH was observed in the outlet reaches during the pre-monsoon period (Table 2). Varthur Lake recorded the maximum pH of 7.9 at the inflows during the premonsoon period and pH range from 7.5 to 7.9 (Table 3). Higher values of EC were observed during the pre-monsoon periods in both Bellandur and Varthur Lakes inflows and outflows and lowest were observed during monsoon.
Highest turbidity values (386 NTU) were recorded in the Bellandur lake inflows during the post-monsoon, and the minimum values were recorded during the monsoon (71 - 79 NTU). Varthur Lake recorded maximum turbidity (325 NTU) values in the inflows during the pre-monsoon period and lowest values during the postmonsoon period. Complete anoxic conditions were observed in Bellandur and Varthur Lakes owing to macrophyte cover and higher organic loads. Higher COD and BOD values were also recorded during the pre-monsoon period in both lakes.
During the entire sampling period, low nitrification was observed as evident from the nitrate values ranging from 0.04 to 1.5 mg/l compared to Amm.-N that ranged from 14 to 65 mg/l. Higher values of Amm.-N (~30 mg/l) were reported by the earlier studies (Chanakya and Sharatchandra, 2008). Nitrate and nitrite concentration constituted an insignificant portion of the dissolved nitrogen pool (< 2.0%). Higher Amm.-N were consistently observed in the inflow regions especially in Bellandur Lake during the post monsoon period indicating an inflow of already decomposing wastewater. TN values were higher during the post monsoon period at the inflows in Bellandur Lake and during the pre-monsoon periods in the inflows of Varthur Lake and are elucidated in Tables 2 and 3 respectively. Ortho phosphates levels ranged from 1.8 - 3.76 mg/l in Bellandur Lake and 1.32 - 3.51 mg/l in Varthur Lake and were comparatively higher in the outlets owing to P re-suspension from anoxic bottom. However, TP values were higher in the inflows compared to the outflows and were ranging from 11 to 37 mg/l in Bellandur Lake and 9e19 mg/l in Varthur Lake.
| Parameters |
Bellandur Lake |
| Pre-monsoon |
Monsoon |
Post-monsoon |
| Inlet |
Outlet |
Inlet |
Outlet |
Inlet |
Outlet |
| pH |
7.9 |
8.2 |
7.4 |
7.8 |
7.7 |
7.8 |
| Temperature (°C) |
27.8 |
27.3 |
24.1 |
23.7 |
24 |
21 |
| Electrical Conductivity (μS/cm) |
1098 |
1057 |
661 |
735 |
980 |
1009 |
| Total Dissolved Solids (mg/l) |
868 |
840 |
526 |
592 |
770 |
808 |
| Turbidity (NTU) |
216 |
96.5 |
71.4 |
79.2 |
386 |
98.9 |
| Dissolved Oxygen (mg/l) |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
| Free CO2(mg/l) |
69 |
21.84 |
48.4 |
61.6 |
64 |
120 |
| COD (mg/l) |
340 |
153 |
266 |
140 |
253 |
122 |
| Filtrable COD (mg/l) |
182 |
102 |
123 |
93 |
133 |
88 |
| BOD (mg/l) |
198 |
74 |
120 |
78 |
170 |
132 |
| Nitrates (mg/l) |
0.05 |
0.04 |
0.76 |
1.63 |
0.93 |
0.67 |
| Amm-N (mg/l) |
54 |
29 |
32 |
19 |
65 |
24 |
| TN (mg/l) |
62 |
44 |
64 |
31 |
69 |
27 |
| Ortho-Phosphates (mg/l) |
2.10 |
1.8 |
3.28 |
3.36 |
2.97 |
3.76 |
| TP (mg/l) |
16 |
12 |
37 |
19 |
28 |
11 |
| Alkalinity (mg/l) |
520 |
300 |
360 |
400 |
260 |
300 |
| Total Hardness (mg/l) |
196 |
204 |
268 |
232 |
292 |
276 |
| Chlorides (mg/l) |
84 |
73 |
82.36 |
85.2 |
86 |
93 |
| Sodium (mg/l) |
54 |
43.82 |
39.7 |
40.4 |
68 |
62 |
| Potassium (mg/l) |
10.4 |
9.72 |
9.1 |
10 |
9.8 |
11.2 |
| ORP (mV) |
-180 |
-150 |
-96 |
-165 |
-245 |
-220 |
Table 2 Seasonal variations in treatment parameters in Bellandur Lake.
| Parameters |
Varthur Lake |
| Pre-monsoon |
Monsoon |
Post-monsoon |
| Inlet |
Outlet |
Inlet |
Outlet |
Inlet |
Outlet |
| pH |
7.6 |
7.76 |
7.85 |
7.53 |
7.9 |
7.5 |
| Temperature (°C) |
28.6 |
26 |
27.7 |
29.1 |
22 |
23 |
| Electrical Conductivity (μS/cm) |
1420 |
1114 |
910 |
897 |
977 |
952 |
| Total Dissolved Solids (mg/l) |
994 |
792 |
720 |
712 |
768 |
755 |
| Turbidity (NTU) |
325 |
108 |
216 |
102 |
161 |
65 |
| Dissolved Oxygen (mg/l) |
0.0 |
0.98 |
0.54 |
0.13 |
0.78 |
0.88 |
| Free CO2(mg/l) |
55 |
12.4 |
40.2 |
21.8 |
14.96 |
8.8 |
| COD (mg/l) |
224 |
192 |
220 |
133 |
188 |
120 |
| Filtrable COD (mg/l) |
146 |
102 |
113 |
86 |
146 |
93 |
| BOD (mg/l) |
172 |
135 |
125 |
78 |
130.47 |
74.9 |
| Nitrates (mg/l) |
0.26 |
0.38 |
1.39 |
1.43 |
0.36 |
0.6 |
| Amm-N (mg/l) |
44 |
14 |
37.5 |
19 |
42 |
22 |
| TN (mg/l) |
49 |
21 |
39 |
22 |
47 |
23 |
| Ortho-Phosphates (mg/l) |
2.66 |
3.51 |
1.72 |
3.04 |
1.32 |
1.72 |
| TP (mg/l) |
18 |
14 |
16.5 |
9 |
19 |
13.7 |
| Alkalinity (mg/l) |
400 |
360 |
360 |
380 |
380 |
330 |
| Total Hardness (mg/l) |
508 |
272 |
248 |
256 |
264 |
288 |
| Chlorides (mg/l) |
446 |
149 |
85.2 |
90.88 |
99.4 |
85.2 |
| Sodium (mg/l) |
1100 |
810 |
42.43 |
42.32 |
220 |
221 |
| Potassium (mg/l) |
80 |
80 |
9.72 |
8.67 |
41.5 |
47 |
| ORP (mV) |
-145 |
-210 |
-13 |
-8 |
-49 |
-178 |
Table 2 Seasonal variations in treatment parameters in Varthur Lake.
3.3. Treatment aspects of lakes
These lakes have been functioning as lagoons or functionally comparable to stabilisation ponds with the thriving algal-bacterial communities that helps in maintaining the overall quality of the water. In the current study, a maximum removal of total COD (~55%); filterable COD (~44%); Net COD (67.7%) and BOD (62.5%) was observed during the pre-monsoon season in Bellandur lake where as filterable COD and BOD removal was better during the post-monsoon period (Table 4 and Fig. S1, Supplementary material) attributable to rapid C uptake and transformations at high temperature with higher insolation through the algal C sequestration route. In the case of Varthur Lake, highest total COD removal (52%) was recorded in monsoon period, where as maximum BOD (~62%); filterable COD (44%) and Net COD (70%) removal took place during the pre-monsoon period and the reasons are similar to the observations in Bellandur lake.
Highet TN removal was observed during postemonsoon period (~61%) in Bellandur Lake attributed to anoxic environments due to large floating macrophytes debrii and floating islands comprising of macrophytes and resuspended sludge on the surface of the lake that drifts towards the outlets. Where as Varthur showed higher N removal during the pre-monsoon peroid (~57%) due to rapid algal uptake. Similarly, the Amm.-N removal was maximum during postmonsoon in Bellandur Lake (~63%) and pre-monsoon in Varthur Lake (~68%) attributable to Amm.-N loses due to increase pH consequent to algal photosynthesis at high light intensities (Weatherell et al., 2003). Higher TP removal was observed in case of Bellandur Lake (~61%) during the post-monsoon peroid, but Varthur Lake showed higher TP removal (~45%) during monsoon. Samples at outlets were having higher values of orthophosphate than inflows attributable to higher mineralisation of organic phosphates as a function of residence time and due to resuspension of P from the lake bottom (Mahapatra et al., 2011a). A higher bacterial removal was observed during the pre-monsoon period in both the lakes due to higher solar illumination, high photosynthesis and consequent development of an increased pH value.
Enormous quantities of terrestrial nutrients gets immobilised in the physicochemical environment and within the biological organisms present in these lakes. Approximately, 100 tonnes BOD/d, 31 tonnes TN/d and 8 tonnes TP/d of enters during pre-monsoon and ~37 tonnes BOD/d, 22 tonnes TN/d and 6 tonnes TP/d of leaves from Bellandur Lake. In monsoon, ~60 tonnes BOD/d, 32 tonnes TN/d and 18.5 tonnes TP/d enters and 39 tonnes COD/d, 15.5 tonnes TN/d and 9.5 tonnes TP/d leaves from Bellandur lake and 85 tonnes BOD/d, 35 tonnes,TN/d and 14 tonnes TP/d entered during post-monsoon period while 66 tonnes BOD/d, 13.5 tonnes TN/d and 5.5 tonnes TP/d leaves the lake.
In the case of Varthur lake, 95 tonnes BOD/d, 27 tonnes TN/d and ~10 tonnesTP/d enters during the pre-monsoon period and ~74 tonnes BOD/d, 11.5 tonnesTN/d and 7.7 tonnes TP/d. During the monsoon peroid, ~69 tonnes BOD/d, 21.4 tonnes TN/d, ~9 tonnes/ d enters Varthur Lake and ~43 tonnes BOD/d, ~12 tonnes TN/d and ~5 tonnes TP/d leaves the system. ~72 tonnes BOD/d, ~26 tonnes TN/d and ~10 tonnes TP/d enters Varthur lake during the postmonsoon peroid and ~41 tonnes BOD/d, ~13 tonnes TN/d and ~7.6 tonnes TP/d leaves the system daily. This provides vital insights for devising methods for recovering nutrients trapped in wastewater fed lakes and further engineer the system for optimal resource recovery and management.
| Water body |
Period |
Percentage Removal |
| BOD |
Total COD |
Fil. COD |
Net .COD (Tot-Filt) |
NO3-N |
NH4-N |
TN |
Ortho Phos. |
TP |
Bacterial cells |
| Bellandur |
Pre-Mon |
62.5 |
55 |
44 |
67.7 |
20 |
46.2 |
29.03 |
14.28 |
25 |
47.72 |
| Mon |
35 |
47.3 |
24.5 |
67.1 |
-53.37 |
40.6 |
51.56 |
-2.38 |
48.64 |
42.3 |
| Post-Mon |
22.4 |
51.7 |
34 |
71.7 |
31.63 |
63.07 |
60.86 |
-21.01 |
60.71 |
25 |
Varthur |
Pre-Mon |
21 |
14 |
30.1 |
-15.3 |
-31.5 |
68.18 |
57.14 |
-24.21 |
22.22 |
57.14 |
| Mon |
37.6 |
39.5 |
23.9 |
56 |
-2.79 |
49.33 |
43.58 |
-43.42 |
45.45 |
31.57 |
| Post-Mon |
42.7 |
36.2 |
36.3 |
35.7 |
-40 |
47.61 |
51.06 |
-23.25 |
27.89 |
18.18 |
Table 4 Physico-chemical characteristics of Bellandur and Varthur Lakes
at inflow and outflow during pre-monsoon (AprileMay), Monsoon (JuneeNovember) and post monsoon (DecembereMarch).
3.4. Algal community dynamics
Over 32 genera of algae were recorded with more than 40 species. Chlorophyceae (13) dominated the algal community, followed by Bacillariophyceae (7), Cynaophyceae (6) and Euglenophyceae (4). Bellandur Lake inlets were dominated by the members of Cynaophyceae (~45%; Merismopedia sp. and Microcystis sp.) followed by Bacillariophyceae (~35%; Gomphonema parvalum) as these species can grow in low light conditions, during the pre-monsoon period. In the middle reaches, when the water is clearer and is free from organic suspenoides, the Chlorophycean members like Chlorella sp. dominated (75%). However, Chlorophyceae (45%) such as Chlorococcum sp. and Monoraphidium sp. dominated outlets. This indicated high photosynthesis by green algal members and requirement of light conditions for rapid photosynthesis (Weatherell et al., 2003). During the monsoon, the Bacillariophycean members dominated the inflows (78%) and Chlorophycean members dominated the middle (78%) and outlets (46%). However, the post monsoon period recorded the highest abundance in the Chlorophycean members (Chlorella sp., Chlorococcum sp. and Monoraphidium sp.) members (62% in middle; 88% in outlets). The inlet region was dominated by diatoms (72%). Biofilms collected from the rock surface, plant surface and sediment layers showed dominance of filamentous algae (Oedogonium sp.) and diatom sp. (Gomphonema sp. and Navicula sp.). The distribution of the different members of phytoplankton at various sampling locations and seasons are presented in Fig. 2. Spatiotemporal variations in algal community's composition are dependent on the nutrient loads, physico-chemical environment and the micro-climatic variables. Solar insolation, precipitation and the wind velocity are crucial in partitioning of the nutrient regimes in the lakes spatially and vertically (Mahpatra et al., 2011b,c).
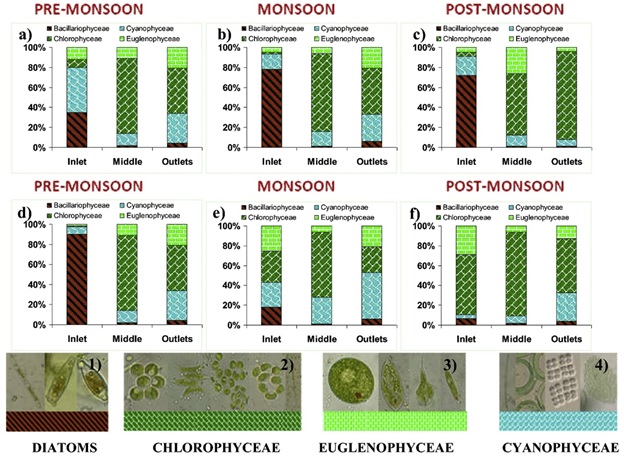 Fig. 2. a), b) and c) represents Bellandur Lake Algal densities at inlet, middle and outlets in various seasons, and d), e) and f) represents Varthur Lake Algal densities at Inlet, middle
Fig. 2. a), b) and c) represents Bellandur Lake Algal densities at inlet, middle and outlets in various seasons, and d), e) and f) represents Varthur Lake Algal densities at Inlet, middle
and Outlets in various seasons. The collage of the algal micrographs below represents: 1) Bacillariophyceae (diatoms); 2) Chlorophyceae; 3) Euglenophyceae and 4) Cyanophyceae.
During pre-monsoon period, in Varthur Lake, diatoms (~90%) dominated the inlet reaches followed by the Chlorophycean members in the middle (75%) and the outfalls (45%). However, during the monsoon period, the Chlorophycean members dominated both at inflow (32%) and middle (66%), while relative abundance of Cyanophyceae (47%) were high near outlets. During the post monsoon, the Cholorophycean members dominated (~80%) in all these three locations. The results showed Scenedesmus sp., Anabaena sp. and Anacystis sp. were predominant near the shorelines while Chlorococcum sp. and Monoraphidium sp. (&lst;10μm) were present during the monsoon season (80%). Algal samples at inlets revealed predominance of Gomphonema sp. and Nitzschia sp. (10 - 38μm) i.e. diatoms during the monsoon, that are succeeded by euglenoides such as Euglena sp. and Phacus sp. (>20μm) in the pre monsoon period. Euglenoides are indicators of organic matter accumulation (Veeresh et al., 2009). During monsoon and post monsoon periods, the algal blooms at Bellandur Lake with higher abundance of Chlorophycean members act as inoculum for Varthur Lake.
Filamentous algae such as Phormidium sp., Oedogonium sp. and Oscillatoria sp. (>30μm) were present near the outlets of the Varthur Lake during monsoon season. Comparative analysis of algal populations in the biofilms revealed a noticeable difference in the community structures at various zones of the Lake. The epilithic (over solid surfaces) algal biofilm consisted of colonies of Stigeoclonium sp. Diatom species such as Gomphonema sp. and Nitzschia sp. were near the inlet regions and species of Chlorophyceae and Euglenophyceae were near outlet regions. These distributions are dependent on the environmental variables such as organic load, nutrients (N and P) and light availability (Mahapatra et al., 2011a). Season-wise investigations indicate that depending upon the wind direction, the extent of growth and movement of the macrophytes together with the nutrient influx, there is a periodic transition from the anaerobic-aerobic (in monsoon) to anaerobic (in summer) and aerobic-anaerobic system (winter/pre monsoon) in the lake. Table S2 (in the Supplementary material) lists the algal composition in studied lakes. Fig. 2 elucidates the season-wise transitions in the algal groups at the various sampling locations. The inflowregions of Varthur Lake suffered from higher anoxia during the pre-monsoon period with only Gomphonema sp. and Nitzschia sp. in shallow regions of the Lake. Inlet regions samples consistently showed a very low count of algal cells in the order of 102-103cells/ml. However, middle portion of the Lake showed an average cell count of 106 - 107cells/ml. The algal cell counts were lower of 103-105 cells/ml in outlet samples, owing to decreased algal abundance with poor interception of sunlight with dense macrophyte cover.
3.5. Bacterio-plankton abundance and distribution
Bacterial growth and community structure are the key biological parameters in wastewater systems. Until recently, bacterial biomass were analysed through either measurements of cell size, usually with image analysis (Blackburn et al., 1998) or epifluorescence microscopy DAPI/Acridine orange (Marie et al., 2017). These are time-consuming techniques and have constraints for continuous and large-scale estimation of bacterial abundance and biomass involving sampling and analysis that needs preservation (Gasol and Giorgio, 2000). In such systems, there is a need to differentially quantify the bacterial and algal biomass to understand the trophic contribution to the purification/treatment process. Flow cytometry (FC) techniques have been used in wastewater microorganisms monitoring especially bacteria for devising and evolving optimal treatment options for urban wastewater treatment. FC has its own advantages in terms of accuracy in cell counting, detecting capability of bacterial species and assessment of live and dead cells (Gasol and Giorgio, 2000). FC aids in the analyses of a large number of cells and recording variability's in cellular characteristics for each cell through several parameters (Shapiro, 2005). In FC, typically 200 to 2000 cells per second circulate (sheath fluid) across beam of a laser and captures the light scattered by each of the particles and the fluorescence emission at different wavelengths generated by the excitation of each particle. FC reduces the time needed for the determination of bacterial abundance, size and activity. In FC usage of various DNA based fluorescent stains, nucleic acid and immunofluorescence probes provide the technique with abilities to discriminate cells on the basis of extent and type of nucleic acids, respiratory enzyme and many other characteristics.
In flow cytometric analysis, three clusters of bacteria were distinguished through normal laser encounter (Fig. 3A-F) with respect to standard beads that were run for dimensionality (Fig. S2, supplementary material). These synthetic beads are used for identifying the potentials that act as markers for the dimension's/ size analysis of bacterio-planktons. Samples from the inlet regions (Fig. S3A) of Bellandur Lake showed a high cell density (107 cells/ ml). However, three different peaks were observed (Fig. S3B) in the middle zone. In samples collected from outlets, only one dominant type of bacteria (Fig. S3C) was observed.
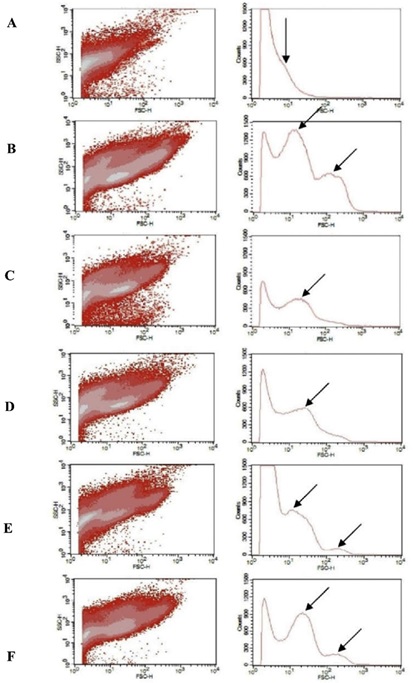 Fig. 3. Flow cytograms for Bellandur Lake (A, B and C) and Varthur Lake (D, E and F).
Fig. 3. Flow cytograms for Bellandur Lake (A, B and C) and Varthur Lake (D, E and F).
Note: The x axis refers to SSC i.e. side scatter that indicates extent of granularity or cellular
complexity and y axis represents FSC i.e.
forward scatter that refers to size of the cells. Arrows indicate different class/groups
of cells gated together on the basis of sizes and
complexities. *A and D represents Inlet; B and E - Middle and C and F - Outlet.
Higher bacterial abundance was observed in the samples of Varthur Lake inlets (shown as single peak in Fig. S3D). Middle regions comprised of large groups of bacteria (Fig. S3E) as highlighted from two peaks indicated by the arrows. Outlet samples comprised mainly several medium sized bacteria represented by a prominent peak depicted in Fig. S3F. The corresponding flow-cytogram (SSC vs. FSC plots) for bacterial abundance and major groups are given in Fig. 3. The scanning electron microscope studies showed different types of bacteria i.e. coccoid, rod-shaped, and curved bacteria (Fig. 4). Among them, the bacillus i.e. rod shaped bacteria were dominant and are indicator organism in wastewater fed systems.
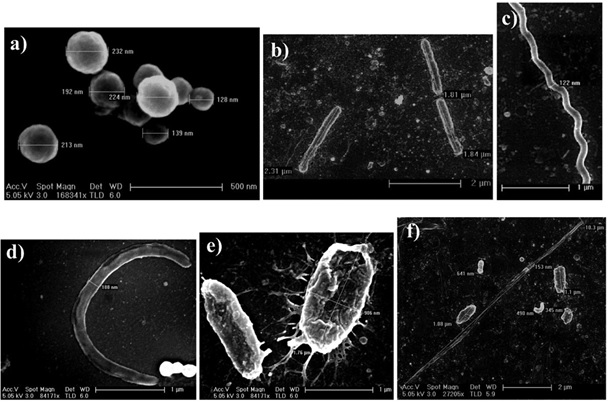 Fig. 4. SEM micrographs of the different shapes of bacteria present in the treatment system a) coccus (avg. size of 200 nm; b) bacillus with numerous lengths (avg. length of 1.8 μm; c) spirillum (width: 120 nm and length ~4 μm) d) C shaped bacteria (avg. length: 3-4 μm and width: ~180 nm) e) activated flocs: projecting out are the filamentous bacteria (average length: 1.76 μm and width: 900 nm) and f) different varieties of bacteria present [i.e. long straight filamentous bacteria (10μm * 150 nm); various kinds of bacillus (sizes: 640 nm to 1.1 μm); comma shaped bacteria (length: ~500 nm).
Fig. 4. SEM micrographs of the different shapes of bacteria present in the treatment system a) coccus (avg. size of 200 nm; b) bacillus with numerous lengths (avg. length of 1.8 μm; c) spirillum (width: 120 nm and length ~4 μm) d) C shaped bacteria (avg. length: 3-4 μm and width: ~180 nm) e) activated flocs: projecting out are the filamentous bacteria (average length: 1.76 μm and width: 900 nm) and f) different varieties of bacteria present [i.e. long straight filamentous bacteria (10μm * 150 nm); various kinds of bacillus (sizes: 640 nm to 1.1 μm); comma shaped bacteria (length: ~500 nm).
3.6. Changes in macrophyte communities and their distribution
The most dominant macrophytes observed were the freefloating water hyacinth (Eichhornia crassipes) and the rooted alligator weed (Alternanthera philoxeroides). Water hyacinth was observed to bloom in the pre monsoon periods during the start of winter mostly in the month of January and then, they grow exponentially in the next three months (JaneApril) and cover almost ~75% of the lake surface. In case of Bellandur Lake, the initial reaches are very shallow and are mostly occupied by the emergent Typha sp. At deeper regions, it is observed the predominance of free floating macrophytes, which are depicted in Fig. S4 (Supplementary material).
As the outlets of Bellandur are clogged with floating islands comprising of rooted macrophytes (water hyacinth is not removed from the Lake), and finally dies and settles down into the water after its growth and death (Battle et al., 2000). New macrophytes utilize this floating mass to form large islands. These floating islands mostly comprise of the lighter weight plants and matured sludge materials that accumulate and adhere to floating mass, because of sludge suspension that progressively compacts, dries and remain as huge floating masses that houses a wide range of macrophytes as Alternanthera philoxeroides, Colocassia, sedges as Cyperus spp. etc. Due to a permanent cover of the floating islands, lakes suffer anoxia due to which no DO was found in the water emerging from the outlets of Bellandur Lake (Mahaptra et al., 2011b,c). These outlet regions therefore, have a highly reducing environment and are associated with odour of H2S.
However, in case of Varthur, the water hyacinth is periodically washed out from the system during monsoon due to high wind velocity and large quantum of water. Sometimes, the local people help in cleaning the clogged outlets to free up water movement during monsoon. Thus, Varthur Lake sheds most of macrophyte islands during monsoon and help to maintain open water surface. During summer, rapidly growing water hyacinth almost entirely covers the surface of the Lake during summer. Upon death, decay and compaction these floating macrophytes are then out-competed by Alternanthera sp. This species is a potential nutrient up-taker grows very rapidly after the death of the water hyacinth plants due to ageing and the weevil attacks. Species like Lemna and Pistia were mostly observed in the Lake edges and in the still and shallow regions. Typha sp. and sedges were observed in inflow regions of the water bodies. Fig. S4 (Supplementary material) shows the different macrophyte species found in these lakes based on nutrient affinity and prevailing environment. It was found that the presence of the floating macrophyte cover resulted in temporary anoxic zones and reduced algal growth, resulting in reduced conditions. Dense macrophyte cover in Varthur Lake has resulted in the absence of light penetration resulting in relatively low algal densities compared to the periods when the outlets are free of macrophytes with a reducing condition of low redox potential (-145 to -210 mV).
3.7. Multivariate analysis
Algal abundance with other physico-chemical parameters linkages were assessed through multivariate statistics. Significant positive correlations (r =0.85; p <0.01) were found between the biomass of Chlorophyceae members with total phosphates (TP). However, the other group of algae as Cyanophyceae were negatively correlated with TP (r = -0.699; p<0.05) which could be due to higher P accumulation capacity of cyanophytes that they use for growth at low P conditions (Kromkamp, 1987). Euglenophyceae members were negatively correlated with total nitrogen (TN) (r= -0.64; p<0.05) whereas Bacillariophycean members aresignificantly negative correlated with filterable COD (r = -0.58; p <0.05) and BOD (r = -0.61; p < 0.05) and are known to prefer nutrient stress environment. On the other hand, if the total algal count (TAC) is examined, the rank correlation results show negative correlations with filterable COD (r = -0.65; p < 0.05), Amm. -N (r = -0.66; p <0.05) and TN (r= -0.73; p< 0.01). Similar results were observed in other studies that showed a bacterial dominance with high concentration of C and N and progressive change as the concentration recedes (Veenstra et al., 1995). Total bacterial counts (TBC) were positively correlated with EC (r= -0.66; p < 0.05), free CO2(r= -0.68; p < 0.05), COD (r= -0.64; p < 0.05), filterable COD (r= -0.69; p< 0.05), Amm. -N (r= -0.88; p <0.01) and total N (r = -0.87; p <0.01) as provided in Table 5. The bacterial predominance at high COD, N and electrolyte concentration are in accordance to the studies by Mahapatra et al. (2013b) at high C and N loads. This shows higher bacterial growth associated with abundance of C and nutrients with high production of CO 2. However, the correlation with EC might be because of higher mineralisation of organics due to faster decomposition with high abundance of bacteria.
Canonical Correspondence Analysis (CCA) showed four distinct regions in the ordinate space characterized by different groups of algae being impacted by specific physicochemical conditions and seasons (Fig. 5). Axis 1 accounted for 60.94% and axis 2 explained 31.73% of the total variation, which jointly represents 92.67% of the total variance. Fig. 5 highlights that parameters such as TP, TN, Amm-N, Turbidity, COD affected the inlets of both lakes (i.e. BM-I, BPOM-I, VPRM-I, VM-I and VPOM-I), showing the abundance of Chlorophycean members. Only the inlets of Bellandur (BPRM-I) were influenced by filterable COD and BOD, Alkalinity, EC and free CO2creating conducive environments for higher bacterial growth and abundance and the highest bacterial counts were observed during the pre-monsoon season. However, the regions near the outlets (BM-O and VPOM-O) were significantly influenced by the inorganic parameters (ionic) as Na, K, Ca and Mg, Chlorides and Nitrate-N where there was predominance of diatom species. Moreover, the outlets (BPOM-O and VM-O) were highly impacted by TDS, pH and temperature pertaining to the growth of Euglenoides.
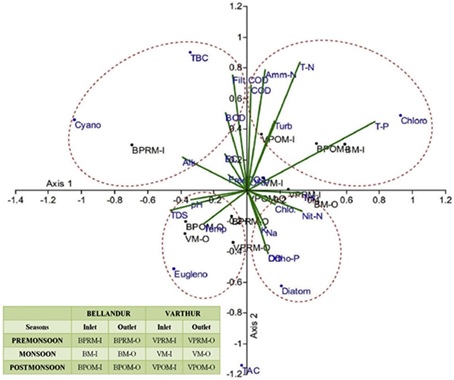 Fig. 5. CCA ordination obtained from algal/bacterial counts and algal relative abundance data and physicochemical variables explaining seasonal variability's (PRM e Pre Monsoon;
M e Monsoon and POM e Post Monsoon) with sampling locations in Bellandur (B) and Varthur (V)Lakes. *Illustration show the ordination of lakes inlet(I)/outlets(O) in different
seasons (represented by points with black dots and black legends as BPRM-I; BM-I; BPOM-I and BPRM-O; BM-O; BPOM-O and VPRM-I; VM-I; VPOM-I and VPRM-O; VM-O; VPOMO)
and physicochemical variables (denoted by green lines and blue legends), and micro-algal genera indicated by blue points and blue legends. (For interpretation of the references
to colour in this figure legend, the reader is referred to the web version of this article.)
Fig. 5. CCA ordination obtained from algal/bacterial counts and algal relative abundance data and physicochemical variables explaining seasonal variability's (PRM e Pre Monsoon;
M e Monsoon and POM e Post Monsoon) with sampling locations in Bellandur (B) and Varthur (V)Lakes. *Illustration show the ordination of lakes inlet(I)/outlets(O) in different
seasons (represented by points with black dots and black legends as BPRM-I; BM-I; BPOM-I and BPRM-O; BM-O; BPOM-O and VPRM-I; VM-I; VPOM-I and VPRM-O; VM-O; VPOMO)
and physicochemical variables (denoted by green lines and blue legends), and micro-algal genera indicated by blue points and blue legends. (For interpretation of the references
to colour in this figure legend, the reader is referred to the web version of this article.)
Bray-Curtis cluster analysis provided the grouping based on spatial similarities of the inlet/outlet seasonality's with different transitions on physico-chemical parameters resulting in changes in biotic activities and communities. The water quality variability in inflows formed four distinct clusters with the outflows especially in the pre-monsoon periods mapping the nutrient concentration with the algal biomass and algal/bacterial abundance (Fig. 6).
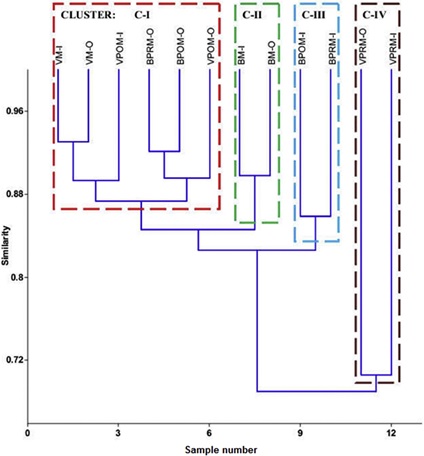 Fig. 6. Dendrogram depicting the similarity in site-seasonality through clustering based on physico-chemical parameters, algal/bacterial abundance and distribution in the studied
lakes.
Fig. 6. Dendrogram depicting the similarity in site-seasonality through clustering based on physico-chemical parameters, algal/bacterial abundance and distribution in the studied
lakes.
Cluster I (87% similarity) comprised of two sub clusters i.e. subcluster-01 (VM-I and VM-O at 93% similarity) with VPOM-I (89.5% similarity) and subcluster-02 (BPRM-O and BPOM-O at 92% similarity) with VPOM-O (89.75% similarity). BM-I and BM-O formed cluster two at a similarity of 90%. Cluster III comprise of BPOM-I and BPRM-I with a similarity of 86% and cluster IV comprised of VPRM-I and VPRM-O at a similarity of 70.25%. The characteristics of VPRM-I and VPRM-O are very different showing significant difference in inlet and outlet characteristics compared to other inflow and outflow characteristics of other seasons as observed by cluster analysis.
| Parameters |
Chlorophyceae |
Euglenophyceae |
Cyanophyceae |
Bacillariophyceae |
Algal Count |
Bacterial Count |
| pH |
-0.53697 |
-0.25773 |
0.27434 |
-0.30773 |
0.038664 |
0.12281 |
| Temperature |
-0.30877 |
0.057471 |
0.13052 |
-0.02853 |
0.15762 |
0.055944 |
| EC |
-0.24211 |
0.23707 |
0.36686 |
-0.10341 |
0.1296 |
0.66434* |
| TDS |
-0.47018 |
0.38793 |
0.38802 |
-0.17829 |
0.32224 |
0.48252 |
| Turbidity |
-0.26714 |
-0.03238 |
-0.11838 |
-0.81263 |
-0.2807 |
0.43783 |
| DO |
-0.17047 |
0.27887 |
-0.11838 |
0.37149 |
0.36491 |
-0.1366 |
| Free CO2 |
0.18246 |
-0.21552 |
-0.10935 |
-0.14976 |
-0.22067 |
0.68531* |
| COD |
0.059649 |
-0.54598 |
-0.40919 |
-0.39223 |
-0.56749 |
0.64336* |
| Filt. COD |
0.082893 |
-0.51817 |
-0.28901 |
-0.57712* |
0.65141* |
0.68542* |
| BOD |
-0.28998 |
0.023389 |
-0.0424 |
-0.6126* |
-0.29123 |
0.55692 |
| Nitrate-N |
0.29474 |
-0.12572 |
-0.34569 |
-0.06062 |
-0.16112 |
-0.40559 |
| Amm-N |
0.11072 |
-0.4192 |
-0.23322 |
-0.56259 |
-0.65965* |
0.87916** |
| TN |
0.31579 |
-0.63578* |
-0.5009 |
-0.30309 |
-0.7285** |
0.87413** |
| Phosphates |
-0.17047 |
0.27887 |
-0.11838 |
0.37149 |
0.36491 |
-0.1366 |
| TP |
0.85237** |
-0.37962* |
-0.69966* |
0.04465 |
-0.28772 |
-0.010508 |
| Alkalinity |
-0.24513 |
-0.28185 |
-0.073215 |
-0.25091 |
-0.26775 |
-0.02832 |
| Tot. Hardness |
0.44562 |
0.43463 |
-0.29631 |
-0.11054 |
0.014011 |
0.12587 |
| Chlorides |
-0.18375 |
0.46664 |
-0.03197 |
-0.42373 |
0.13404 |
-0.21127 |
| Na |
-0.03158 |
0.5388 |
0.17285 |
-0.36014 |
-0.20315 |
0.06993 |
| k |
-0.01056 |
0.38209 |
0.14336 |
-0.29163 |
- 0.17223 |
-0.02456 |
| ORP |
0.035088 |
-0.3592 |
-0.10935 |
0.032092 |
-0.21016 |
-0.29371 |
Table 5 Spearman's rank correlation between physico-chemical variables and groups of algae growing in the Lakes during the study period.
3.8. Nutrient dynamics and budgeting
The monsoon and the post-monsoon witnessed a significant quantity of BOD removal inferring higher organic matter degradation and uptake. High BOD removal is noticed at locations with open lake surface (without macrophytes cover) with active aerobic environments, which is also evident from high DO concentrations in the Lake. Higher DO levels were due to algal photosynthesises and surface aeration that lowered BOD. Compared to this, thick macrophyte cover during late winter and summer months resulted in poor BOD removal. The lake has been functioning in tandem as an anaerobiceaerobic lagoon imparting a satisfactory treatment in the lake. Therefore, understanding the biotic community succession with the wetland dynamics is essential to arrive at appropriate treatment strategies and devising location specific constructed wetland that enhances the sustainable water treatment and nutrient bioremediation. This also helps in replicating similar strategies in the emerging cities and towns in India and many developing nations across tropics in addressing the water scarce situations.
Approximately ~200 tons of BOD enters the lake daily. Considering higher efficiencies of anaerobic-aerobic lagoon systems, a larger loading and conversion are plausible. De-silting would increase of storage volume, while enhancing the water residence time and treatment. Matching the influent concentrations to the assimilative capacity of the lakes will enhance efficacy of the lake systems with a lively biodiversity that maintains the integrity of the lake. The differential activity of the various communities in the lakes depends upon the nature and type of the wastewater fed into the systems and is mostly driven by various environmental factors such as solar insolation, temperature, pressure, wind speed and rainfall.
About 82 tonnes BOD/d and 78 tonnes, BOD/d enters Bellandur and Varthur Lakes respectively (comparable to typical lagoons). On the other hand, when one considers the maximum potential of the anaerobiceaerobic systems, higher loading rates and higher conversionrates are possible. There is, thus, a need to further examine the potential for higher quality of water at the outlet to enable the recycling and reuse of water in the future upon de-silting and increasing the residence time or reducing the loading. In order to make this more sustainable, the extent of the harvest and the reuse of plant nutrients for the system need to be examined. The contribution of macrophytes and phytoplankton in removing nutrients in these sewage-enriched systems varies with the nature of the effluent and the age of the wetland, in addition to other environmental factors like sunlight, temperature, wind and precipitation.
3.9. Nutrient-integrated treatment efficiencies
Crucial treatment parameters are indicative of the remediation performance of urban wetlands. The important factors that influence the treatment ability of the surface water bodies are influent wastewater concentrations, loading rates, microbial community, and the water residence time. The treatment parameters at different phases of treatment determine the degradation of organic matter and transformations, nutrients uptake, and changes in the physic-chemical environment on microbial growth. Concentrations of influent wastewater, volumetric loading, microbial composition, water residence time are the key factors that drive treatment competence. Thus, formulating appropriate treatment efficiency indices are key to the overall efficiency, which helps in evolving future management plans for effective decisions making. Many treatment parameters such as suspended solids, COD, BOD and NH4-N removal efficiencies have been considered for assessing the efficiency of the treatment systems (Colmenarejo et al., 2006). Inegrated treatment efficiency in the present case has been devised taking into consideration all the crucial parameters from the treatment perspectives that focus on clarity of the water, C, nutrient content with bacterial counts and equal weightages have been considered for these parameters. Such Indices help in assessing the treatment levels to cater to an accepted water quality level to be used for any purpose. This helps in deciding additional treatment options for complete treatment of wastewater. Pond based systems in tropics have been used for polishing and maintaining water quality for the end use after treatment and also cleans of bacteria (Jimenez, 2007). Such systems on an average show efficiency close to 90%. In the present study the treatment interacted efficiency is computed as per equation (2).
TIE = [ETUR + ECOD + EN + Ep + EBAC]/5.......................(2)
where
TIE : Treatment Integrated Efficiency (in percentage),
ETUR : turbidity removal efficiency (in percentage),
ECOD : COD removal efficiency (in percentage),
EN : BOD5 removal efficiency (in percentage),
Ep : Nitrogen removal efficiency (in percentage), and
EBAC : Phosphorus removal efficiency (in percentage).
TIE values for Bellandur and Varthur Lakes were 64.85 and 63.19% respectively indicating poor treatment efficiencies due to enormous C and nutrients loading beyond their assimilative capabilities and lower water residence time. This emphasises the need for checking the volume of C and nutrient loads to cope up to their treatment potential. This necessitates
a) increasing the oxidative surfaces by regular clearing invasive floating macrophytes (except functionally active floating islands of macrophytes) that hinders sunlight penetration and natural air diffusion in the system,
b) provisions for increasing the water residence time by wet dredging for better treatment (which also increases the storage capacity),
c) regulating the quality and quantity of wastewater loads by allowing treatments in the upstream of respective wetlands.
|










