Dr. T.V. Ramachandra
Centre for Sustainable Technologies, Centre for infrastructure, Sustainable Transportation and Urban Planning (CiSTUP), Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. E-mail : cestvr@ces.iisc.ac.in Tel: 91-080-22933099/23600985, Fax: 91-080-23601428/23600085 Web: http://ces.iisc.ac.in/energy
Sudarshan P. Bhat
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. E-mail: sudarshan@ces.iisc.ac.in
Asulabha K.S.
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. E-mail: asulabha@ces.iisc.ac.in
Sincy
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. E-mail: sincy@ces.iisc.ac.in
Citation: Ramachandra T V, Sudarshan P Bhat, Aslulabha K S and Sincy V, 2014. Agony of Chikkabettahalli lake, Vidyaranyapura, Bruhat Bangalore, ETR 83, Energy & Wetlands Research Group, CES, Indian Institute of Science , Bangalore
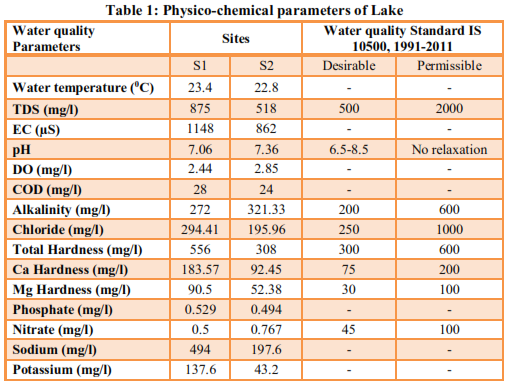
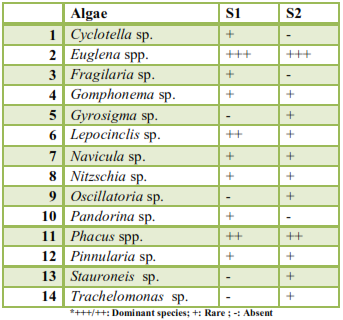
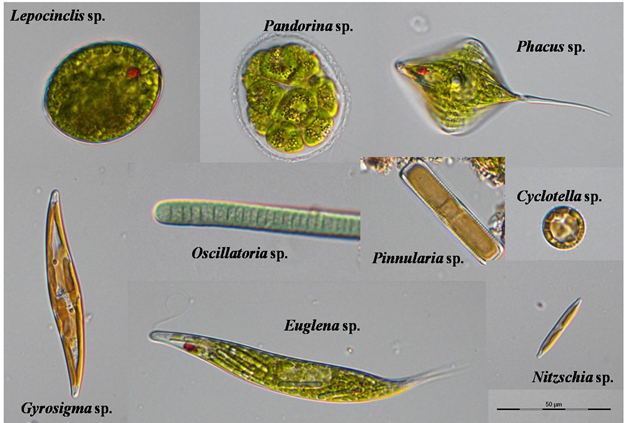
| ||||||||||||||||||||||||