|
Results and Discussion
Variations in Physico-chemical Parameters of Lakes
The lakes monitored in this study varied in their
physico-chemical characteristics as these lakes receive
untreated sewage or pollutants. An alteration of the physical,
chemical, and biological properties of water happens due to
pollution with the sustained inflow of untreated or partially
treated wastewater from domestic and industrial sources
(Udhayakumar et al., 2016). The changes in water
quality can alter phytoplankton community structure and
distribution in lakes. The discussions on physico-chemical
parameters analyzed are elaborated below:
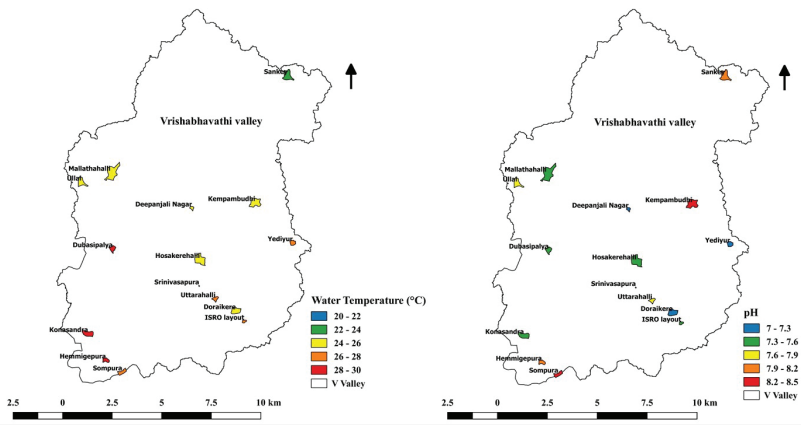
Water Temperature and pH
Temperature strongly influences the chemical composition of
cells, uptake of nutrients, uptake of carbon-dioxide, and growth
rate of phytoplankton (Singh and Singh, 2015;
Brönmark and Hansson, 2002). Water temperature varied
between
Biodiversity Challenges: A Way Forward
Figure 10.2: Variation of Water Temperature and pH in Lakes
of Bangalore.23.9 oC to 29.7 oC in monitored
lakes during the current study (Figure 10.2). pH indicates hydrogen
ion concentration in lake water (Kumar and Mahajan, 2020) and is
generally governed by the equilibrium between carbon-dioxide,
carbonate, and bicarbonate ions. Here, alkaline conditions prevailed
in lakes, which ranged from 7.01 – 8.5. This range of pH is suitable
for phytoplankton growth. During photosynthetic activity by
phytoplankton, pH increases due to the consumption of
carbon-dioxide.
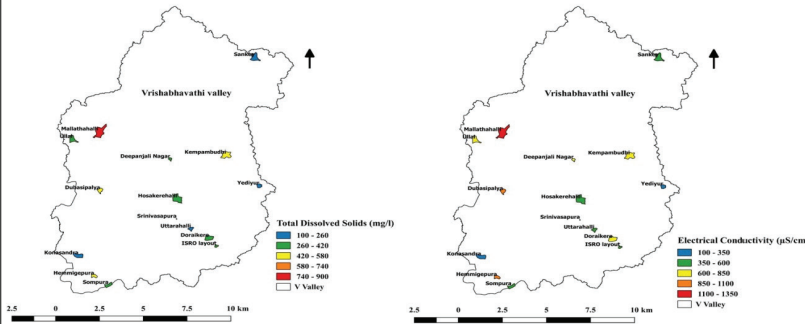
Total Dissolved Solids (TDS) and Electrical Conductivity
(EC)
The total dissolved solids (TDS) include inorganic salts and
small amounts of organic matter in water. The concentration of
total dissolved solids ranged from
152.5 mg/L to 883.42 mg/L in lakes (Figure 10.3). The variations
in TDS occur naturally from rocks and soil as well as from
anthropogenic sources through sewage, industrial effluent
discharges, and urban run-off (Ntengwe, 2006). The conductivity
is a measure of the capacity of water to conduct an electric
current. The value of electrical conductivity ranged from 306.67
µS/cm to 1303.67 µS/ cm (Figure 10.3). EC depends on the type,
quantity, and ionic state of dissolved substances
(electrolytes), thus, indicates the mineral content of water (Le
et al., 2017). Mallathahalli lake had the highest level
of TDS and EC, hence shows ionic pollution.
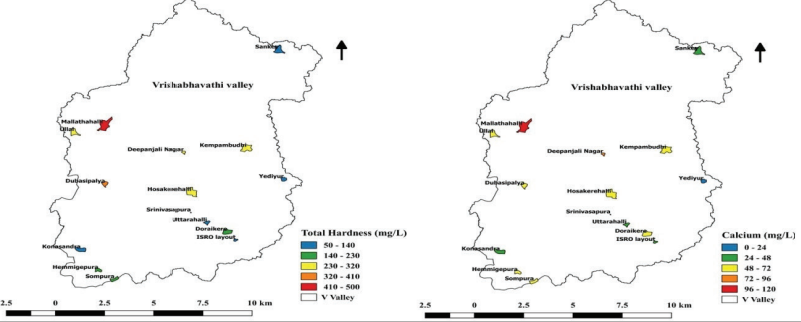
Total Hardness, Calcium, and Magnesium
Hard water is not suitable for domestic and industrial purposes.
The cations that impart total hardness to water are calcium,
magnesium, iron, strontium, and manganese. The total hardness,
calcium, and magnesium varied among sampled lakes and ranged
from 78.67 mg/L- 463.5 mg/L, 23.51 mg/L - 117.9 mg/L, and 4.85
mg/L - 41.09 mg/L,respectively (Figure 10.4). Mallathahalli lake
had the highest levels of total hardness, calcium, and
magnesium, which indicates ionic pollution.
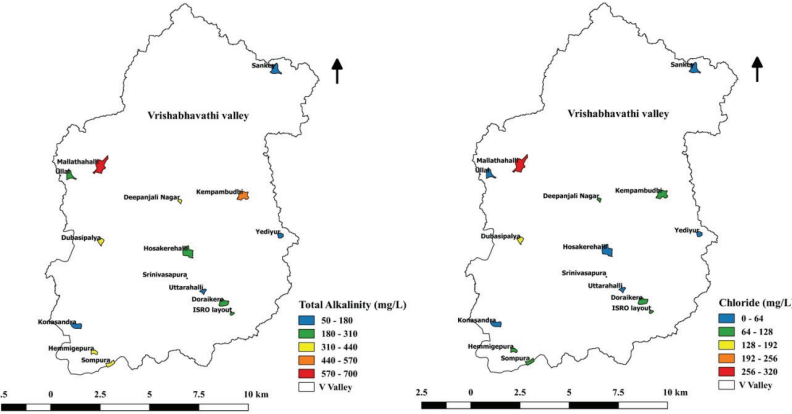
Total Alkalinity and Chloride
Total alkalinity is determined by the concentration of hydroxide,
carbonate, and bicarbonate in water. Thus, total alkalinity
influences the composition and abundance of phytoplankton (Bose
et al., 2019).
Mallathahalli lake receives a lot of untreated domestic and
industrial sewage, so profuse growth of phytoplankton was
observed that increased the alkalinity of lake water. In
addition, the discharge of domestic and industrial wastes
increases chloride levels in lake water. The concentration of
total alkalinity and chloride in lakes ranged from 81.33 mg/L -
684.33mg/L and 26.74 mg/L - 305.89 mg/L, respectively (Figure
10.5).
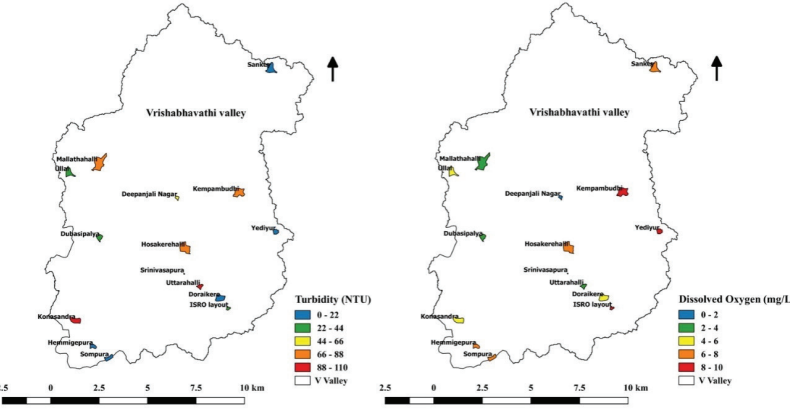
Turbidity and Dissolved Oxygen (DO)
Turbidity in water occurs mainly due to particles of clay, silt,
organic particles, inorganic matter, micro-organisms, and algae
bloom. The range of turbidity in monitored lakes was found as
5.77 NTU - 103.51 NTU (Figure 10.6). Under
Biodiversity Challenges: A Way Forward
Figure 10.3: Level of Total Dissolved Solids and Electrical
Conductivity in Lakes.
Figure 10.4: Concentration of Total Hardness and Calciumin
Lakes.
Biodiversity Challenges: A Way Forward
Figure 10.5: Concentration of Total Alkalinity and Chloride
in Lakes.turbid conditions, only less light can
penetrate through the water column to the lake bottom. This results
in inhibition of the growth of phytoplankton and lower
photosynthetic activity (Yang et al., 2012). This may, in
turn, decrease the dissolved oxygen level in lake water. The
dissolved oxygen levels in lakes ranged from 0.81 mg/L - 9.84 mg/L
(Figure 10.6). Dissolved oxygen affects the various physico-
chemical attributes and biological processes in lake water
(Ramachandra et al., 2014).
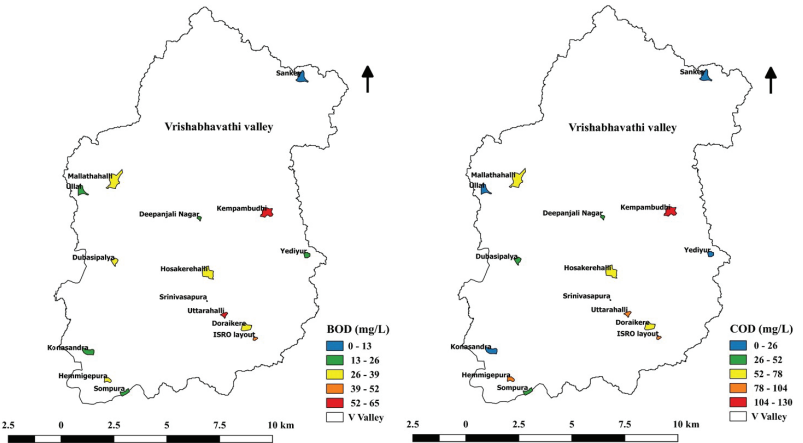
Biochemical Oxygen Demand (BOD) and Chemical Oxygen Demand
(COD)
Biochemical oxygen demand (BOD) determines the amount of oxygen
required by aerobic micro-organisms to stabilize the organic
matter (Avvannavar and Shrihari, 2008). High COD concentrations
indicate pollution related to untreated domestic sewage and
industrial effluents. The BOD and COD of monitored lakes ranged
between 12.2 mg/L - 60.97 mg/L and 21.33 mg/L - 128 mg/L,
respectively (Figure 10.7). The higher levels of BOD and COD
show the level of pollution, which will increase the oxygen
demand in lakes. Among the sampled lakes, Kempambudhi lake had
more elevated BOD and COD, indicating organic pollution.
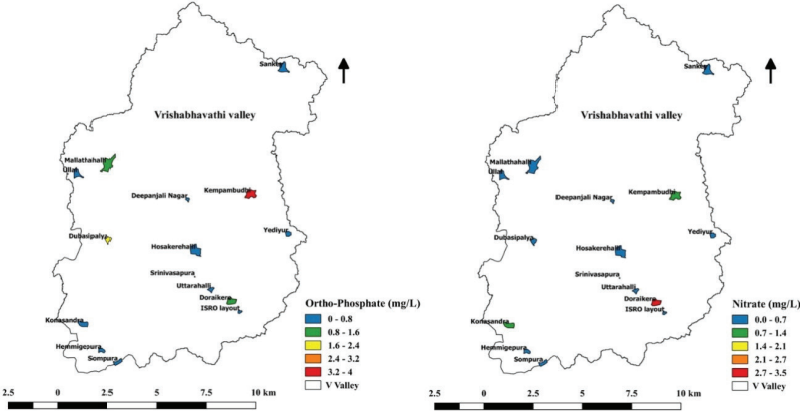
Nitrate and Ortho-phosphate
Nutrients such as nitrogen and phosphorus are the limiting
factors for phytoplankton growth. In the present study, nitrate
and ortho-phosphate ranged between 0.01 mg/L - 3.1 mg/L and
0.077 mg/L - 3.701 mg/L, respectively (Figure 10.8). The
excessive amount of nitrogen and phosphorus can stimulate
eutrophic conditions (He et al., 2011). The discharge
of phosphorus into aquatic ecosystems results in eutrophication,
whichhas enhanced the overgrowth of algae and macrophytes
(Çelekli and Sahin, 2021). Kempambudhi lake was found to be rich
in nitrate and ortho-phosphate (OP), which indicates nutrient
pollution.
The study reveals that (i) Mallathahalli lake had higher levels
of TDS, EC, total hardness, calcium, magnesium, chloride, total
alkalinity, (ii) Kempambudhi lake with high pH, BOD, COD, and OP
show organic and nutrient pollution, and (iii)Yediyur lake had
less ionic contents such as TDS, total hardness, calcium,
magnesium, and COD. Thus, the monitored lakes are polluted as
they receive untreated domestic waste, agricultural residues,
and industrial effluents. This may threaten biodiversity, reduce
the aesthetic and recreational value of lakes.
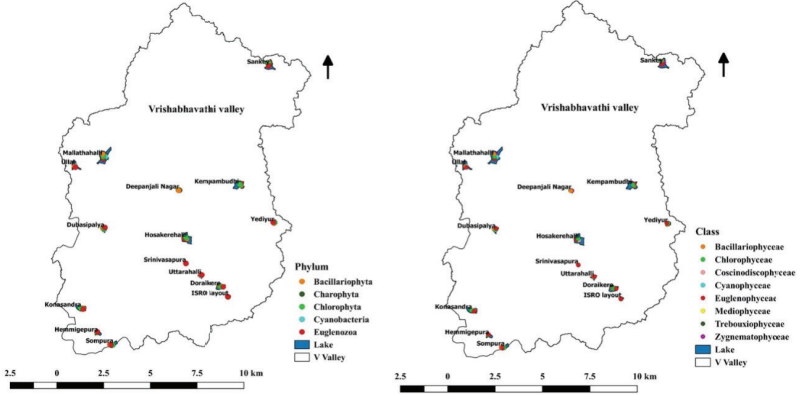
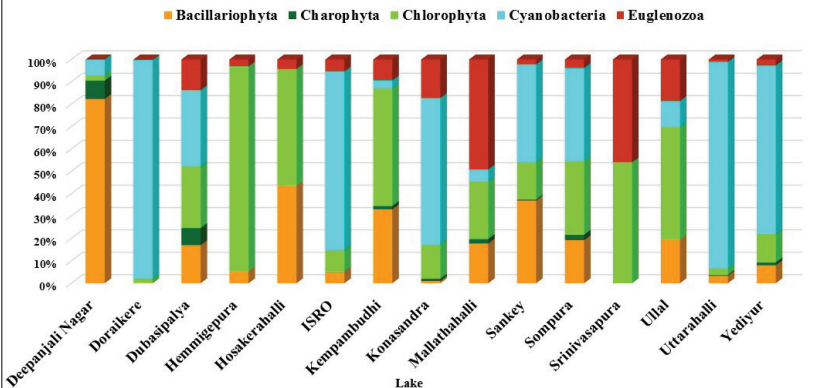
Phytoplankton Community in Lakes
The present study covers 15 lakes in Vrishabhavathi valley of
Bangalore, which recorded a total of 58 genera of phytoplankton
belonging to 5 phyla, 9 class, 24 orders, and 41 families were
identified (Figure 10.9). The fifty-eight species of
phytoplankton were represented by five major groups, namely,
Chlorophyta (25 species), Bacillariophyta (12 species),
Cyanobacteria (12 species), Charophyta (5 species), and
Euglenozoa (4 species).
Biodiversity Challenges: A Way Forward
Figure 10.6: Turbidity and Dissolved Oxygen Levels in
Lakes.
Figure 10.7: Variation of BOD and COD in Lakes.
Biodiversity Challenges: A Way Forward
Figure 10.8: Concentration of Nitrate and Ortho-phosphate in
Lakes.Diversity of Phytoplankton in Lakes of
Bangalore, Karnataka, India
Figure 10.9: Phytoplankton Composition (Phylum and Class) in
Lakes of Bangalore.Cyanobacteria, commonly known
as blue-green algae, are photosynthetic prokaryotes that form algal
blooms in water (Elliott, 2012). Cyanobacteria are represented by
species such as Anabaena sp., Aphanocapsa sp.,
Chroococcus sp., Coelosphaerium sp., Cylindrospermopsis sp.,
Gloeocapsa sp., Merismopedia sp.,
Microcystis sp., Oscillatoria sp.,
Phormidium sp., Planktothrix sp. and
Spirulina sp. Cyanobacteria, mainly Microcystis sp.was
dominant and formed surface blooms in Doraikere, Uttarahalli, ISRO
layout, and Yediyur lakes in Vrishabhavathi valley, Greater
Bangalore, Karnataka (Figure 10.10). A total of 25 species of
Chlorophyta was recorded in this study, such as
Actinastrum sp., Ankistrodesmus sp., Asterococcus sp.,
Botryococcus sp., Chlamydomonas sp.,
Chlorella sp., Chlorococcum sp.,
Coelastrum sp., Crucigenia sp.,
Desmodesmus sp., Dictyosphaerium sp., Golenkinia sp.,
Kirchneriella sp., Micractinium sp., Monoraphidium sp.,
Oocystis sp., Pandorina sp., Pediastrum sp.,
Scenedesmus spp., Schroederia sp., Stichococcus sp.,
Stigeoclonium sp., Tetraedron sp. and Tetrastrum sp.
Both pennate and centric forms of Bacillariophyta (or diatoms) are
present lakes inVrishabhavathi valley,
Bangalore.Achnanthes sp., Amphora sp., Cyclotella sp.,
Fragilaria sp., Gomphonema sp., Melosira sp.,
Navicula sp., Nitzschia sp., Pinnularia sp.,
Stauroneis sp., Surirella sp. and
Synedra sp. belonging to Bacillariophyta was recorded.
A total of four species under Charophyta were recorded, such as
Closterium sp., Cosmarium sp.,
Euastrum sp., Spirogyra sp. and Staurastrum sp.
in the present investigation. Euglenozoa is represented by
species such as Euglena spp., Lepocinclis sp.,
Phacus sp. and Trachelomonas sp.
Euglena sp. is mixotrophic algae, which can grow faster
in wastewater because they can utilize organic matter as an
energy source (Mahapatra et al., 2013).
Diversity Indices
Various diversity measures were used to estimate the
phytoplankton diversity of lakes in Vrishabhavathi valley,
Bangalore. Shannon-Wiener index ranged from
0.16 -2.54. Shannon and Wiener index value of 0.0-1.0 indicates
heavy pollution, 1.0-2.0 indicates moderate pollution, 2.0-3.0
indicates light pollution, and 3.0-
4.5 indicates slight pollution. The species diversity indices
decrease in polluted waters (Shanthala et al., 2009).
Thus, Doraikere (2.54) is heavily polluted with less species
diversity. Simpson index ranged from 0.045 (Doraikere) -0.882
(Sompura, Mallathahalli). Pielou evenness index reveals an even
distribution of individuals among varied species (Miao et
al., 2019). The evenness index ranges from near 0 (low
evenness) to 1 (dominance). Evenness index ranged from 0.07
(Uttarahalli)
– 0.80 (Deepanjali Nagar, Srinivasapura). Menhinick index ranged
from 0.26 (Doraikere) – 1.59 (Hemmigepura) in this study.
Margalef species richness index measures the number of different
species in each sample (Ray et al., 2021). Margalef
index ranged from 0.72 – 4.77 as Dubasipalya lake has the
highest taxa whereas Srinivasapura has the lowest taxa and
individuals. A high value of richness index indicates healthy
status, while a low value indicates the unhealthy and
contaminated status (Hussien and Zaghloul, 2017). The
Berger-Parker index gives the measure
Figure 10.10: Phytoplankton Composition in Lakes of
Bangalore.of the most abundant species (Berger
and Parker, 1970). Berger-Parker index in this study ranged from
0.21 (Deepanjali Nagar) -0.98 (Doraikere).
Correlation between Water Quality Parameters and
Phytoplankton
Pearson correlation analysis is used to measure the interrelation
and extent of associations among the water quality parameters
and algal phyla. Bacillariophyta negatively correlated with BOD
(r = -0.31) and COD (r = -0.38). Bacillariophyta responds
rapidly to organic and nutrient contamination (Saxena et
al., 2020). Charophyta positively correlated with water
temperature (r = 0.41), electrical conductivity (r = 0.36),
magnesium (r = 0.40), ortho-phosphate (r = 0.31) and
Bacillariophyta (r = 0.57). Chlorophyta positively correlated
with water temperature (r = 0.38), Charophyta (r = 0.68) and
Bacillariophyta (r = 0.90). A positive correlation was found
between temperature and phytoplankton such as Charophyta and
Chlorophyta (Gogoi et al., 2019) as temperature plays
an important role in growth and reproduction of phytoplankton.
Cyanobacteria negatively correlated with pH (r = -0.45), total
hardness (r = -0.44), calcium (r = -0.39), magnesium (r= -0.46),
total alkalinity (r = -0.49) and positively correlated with
nitrate (r = 0.63). A similar pattern of positive correlation
recorded between Cyanophytes with nitrate, and inverse
correlation with alkalinity, pH, total hardness, calcium and
magnesium (Mishra et al., 2019). In Baiyangdian lake,
Cyanophyta and Chlorophyta became dominant because of increased
organic matter from industrial wastewater and domestic sewage
(Wang et al., 2013). Euglenozoa positively correlated
with TDS (r = 0.49), EC (r = 0.59), total hardness (r = 0.49),
calcium (r = 0.36), magnesium (r = 0.61), chloride (r = 0.58),
total alkalinity (r = 0.43), ortho-phosphate (r = 0.31),
Bacillariophyta (r = 0.39), Charophyta (r = 0.79), Chlorophyta
(r = 0.72) and negatively correlated with DO (r = -0.30). The
dominance of Euglenophyta indicates organic pollution, water
pollution, and eutrophic condition of water body (Shams and
Shamsabadi, 2019).
Relationship between Phytoplankton Community and Physico-
chemical Factors
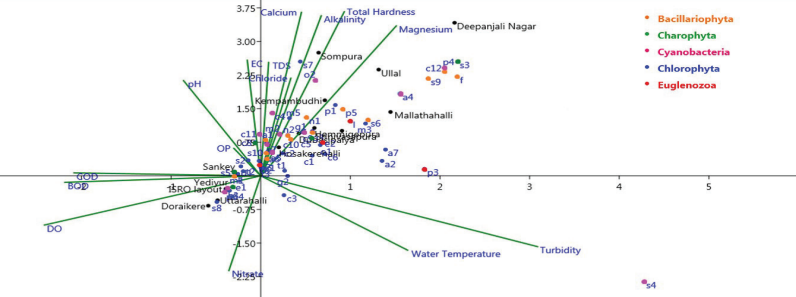
Canonical correspondence analysis (CCA) was computed with
phytoplankton composition data (58 genera), 15 physico-chemical
parameters across 15 lakes during the study period to evaluate
the role of environmental variables in structuring phytoplankton
communities. For CCA, 15 physico-chemical parameters, namely:
water temperature, pH, conductivity, total alkalinity, dissolved
oxygen, nitrate, ortho-phosphate, turbidity, TDS, BOD, COD,
chloride, calcium, magnesium, and total hardness,were selected.
Different species of phytoplankton showed varied responses to
physico-chemical parameters. The length of the arrow in CCA
ordination indicates the relative importance of environmental
variables. Eigenvalues of the first two axes are 0.58 and
0.48,which explain 34.97 per cent of the variance of
phytoplankton and environmental data. Axis 1 has strong positive
loadings of
turbidity. Thus, Spirulina sp., Actinastrum sp.,
Asterococcus sp., Micractinium sp. and Phacus sp.
are positively correlated with the turbidity. Turbidity of
freshwater lakes increases due to the influx of urban run-off,
silt, sand, and organic matter (Nandigam et al., 2016).
This decreases the phytoplankton diversity and increases the
dominance of Spirulina sp. as in Konasandra lake.
Axis 2 has strong positive loadings of total hardness, calcium,
magnesium, and total alkalinity. Total hardness, calcium,
magnesium, and total alkalinity are found in lakes such as
Deepanjali Nagar, Hemmigepura, Sompura, Kempambudhi,
Srinivasapura, Ullal, Doraikere, Dubasipalya, Hosakerehalli, and
Uttarahalli. Therefore, the growth of species such as Anabaena sp.,
Chroococcus sp., Cymbella sp.,
Fragilaria sp., Gomphonema sp., Lepocinclis sp.,
Monoraphidium sp., Navicula sp., Oscillatoria sp.,
Pandorina sp., Phormidium sp.,
Pinnularia sp., Spirogyra sp.,
Stauroneis sp., Stichococcus sp. and Surirella sp.
are positively dependent on the parameters like total hardness,
calcium, magnesium, and total alkalinity. Magnesium is crucial
for chlorophyll synthesis and serves as a limiting factor for
phytoplankton growth and reproduction (Narchonai et
al., 2019). The remaining species did not show a
relation with the selected variables. Phytoplankton species such
as Amphora sp., Aphanocapsa sp., Botryococcus sp.,
Chlorococcum sp., Desmodesmus sp., Dictyosphaerium sp.,
Euastrum sp., Golenkinia sp., Kirchneriella sp.,
Melosira sp., Microcystis sp.,
Pediastrum sp., Planktothrix sp., Scenedesmus spp.,
Schroederia sp., Staurastrum sp., Stigeoclonium sp.,
Tetraedron sp., Tetrastrum sp. and Trachelomonas sp.
found nearer to the center of CCA ordination has a broad range
of tolerance to the variations in physico-chemical parameters in
lakes. Anabaena sp., Merismopedia sp., Microcystis sp.
and Oscillatoria sp. indicate eutrophic lake conditions
(Yilmaz et al., 2018).
Palmer Index
The phytoplankton community composition alters quickly due to
changes in the physico-chemical characteristics of water. For
example, the presence of varied species of phytoplankton can
indicate the quality of water. For example, Oscillatoria and
Euglena are indicators of dirty water, while Pediastrum
indicates clean water (Kantachote et al., 2009).
Thus,phytoplankton communities serve as indicators of pollution
in an aquatic ecosystem. The Palmer index is a rapid, efficient,
and cost- effective tool that reveals the water quality status
or water pollution in freshwater bodies (Salem et al., 2017).
Palmer prepared a list of algal genera and species that are
tolerant to varying levels of organic pollution (Vishal and
Meeta, 2020). Palmer index accounts for the abundant pollution
tolerant phytoplankton taxa and categorizes water bodies based
on the pollution status. The palmer index value of 20 or more
confirms high organic pollution. A value of 15-20 indicates
probable organic pollution. The values between 10-15 and 0-10
show moderate pollution and lack of organic pollution,
respectively (Jose and Kumar, 2011).
Biodiversity Challenges: A Way Forward
Figure 10.11: Canonical Correspondence Analysis (CCA)
Ordination Diagram Showing Scatter Plot for Phytoplankton, Lakes
and Physico-chemical Parameters (Arrows)(Abbreviations:
a1: Achnanthes sp.; a2: Actinastrum sp.; a3: Amphora sp.;
a4: Anabaena sp.; a5: Ankistrodesmus sp.; a6: Aphanocapsa sp.;
a7: Asterococcus sp.; b: Botryococcus sp.; c1:
Chlamydomonas sp.; c2: Chlorella sp.; c3: Chlorococcum sp.;
c4: Chroococcus sp.; c5: Closterium sp.; c6: Coelastrum sp.;
c7: Coelosphaerium sp.; c8: Cosmarium sp.; c9:
Crucigenia sp.; c10: Cyclotella sp.; c11: Cylindrospermopsis sp.;
c12: Cymbella sp.; d1: Desmodesmus sp.; d2: Dictyosphaerium sp.;
e1: Euastrum sp.; e2: Euglena spp.; f- Fragilaria sp.;
g1: Gloeocapsa sp.; g2: Golenkinia sp.; g3: Gomphonema sp.;
k: Kirchneriella sp.; l: Lepocinclis sp.; m1: Melosira sp.;
m2: Merismopedia sp.; m3: Micractinium sp.; m4:
Microcystis sp.; m5: Monoraphidium sp.; n1: Navicula sp.;
n2: Nitzschia sp.; o1: Oocystis sp.; o2: Oscillatoria sp.;p1:
Pandorina sp.; p2: Pediastrum sp.; p3:
Phacus sp.; p4: Phormidium sp.; p5:
Pinnularia sp.; p6: Planktothrix sp.; s1: Scenedesmus spp.;
s2: Schroederia sp.; s3: Spirogyra sp.; s4: Spirulina sp.;
s5: Staurastrum sp.; s6: Stauroneis sp.; s7: Stichococcussp.;
s8: Stigeoclonium sp.; s9: Surirella sp.; s10-
Synedra sp.; t1: Tetraedron sp.; t2: Tetrastrum sp.
and t3: Trachelomonas sp.).
According to Palmer index (Table 10.1), Doraikere, Deepanjali
Nagar, Srinivasapura, Hosakerahalli lakes had moderate pollution
as the Palmer index for these lakes was 10 -15.On the other
hand, lakes such as ISRO Layout, Uttarahalli, Sankey, Yediyur,
Sompura, Ullal, Hemmigepura, Konasandra, Dubasipalya,
Mallathahalli, and Kempambudhi had high organic pollution (with
Palmer index of 20 or more) in the present study. The dominance
of species such as Chlorella, Oscillatoria, Pediastrum,
Scenedesmus, Nitzschia, Melosira, Gomphonema, Navicula, Euglena,
etc., indicates of organic pollution in water bodies
(Kshirsagar, 2013; Parvez et al., 2019). The Palmer
algal genera pollution index was in the range of 15 -24 in
Vaigai river in Madurai (Noel and Rajan, 2015), while 5 - 28.6
in Chulband River in Maharashtra,which indicate organic
pollution (Shahare, 2017). Palmer pollution index revealed that
Sidi Abderrahmane is an oligotrophic or mesotrophic reservoir
(Belokda et al., 2019). Any alteration in the water
quality of the aquatic ecosystem will alter the phytoplankton
community structure. Only pollution tolerant species can thrive
well in polluted waters, whereas sensitive species disappear, as
they cannot survive in polluted water. Thus, phytoplankton
serves as a bioindicator for assessing the extent of pollution
of water bodies (Abdulwahid, 2016).
Table 10.1: Palmer Pollution Index of Lakes Based on
algal genus
Lake |
Index Level |
Remark |
ISRO |
21 |
High organic pollution |
Uttarahalli |
27 |
High organic pollution |
Sankey |
25 |
High organic pollution |
Yediyur |
20 |
High organic pollution |
Doraikere |
14 |
Moderate pollution |
Sompura |
35 |
High organic pollution |
Ullal |
35 |
High organic pollution |
Hemmigepura |
26 |
High organic pollution |
Konasandra |
30 |
High organic pollution |
Dubasipalya |
39 |
High organic pollution |
Mallathahalli |
23 |
High organic pollution |
Deepanjali Nagar |
10 |
Moderate pollution |
Srinivasapura |
10 |
Moderate pollution |
Hosakerehalli |
12 |
Moderate pollution |
Kempambudhi |
25 |
High organic pollution |
The physico-chemical parameters are the major factors controlling
the structure and composition of phytoplankton in freshwater
ecosystems. Phytoplanktons are an important component of an
aquatic ecosystem and are responsible for primary productivity.
The development of phytoplankton was favored by higher
concentrations of nutrients and organic matter (Devercelli
and
O’Farrell, 2013). Water quality parameters such as temperature,
pH, DO, EC, BOD, COD, phosphate, and nitrate influenced
phytoplankton species composition and distribution (Cheraghpour
et al., 2013). Phytoplankton grows well in nutrient-
rich water and removes BOD, COD, and nutrients while providing
oxygen to other aquatic organisms (Kiran et al., 2016).
The abundance of Chlamydomonas sp.,
Cyclotella sp. and Euglenophyta indicates the excessive
availability of N and P as these species are organic-tolerant
(Ghobara and Salem, 2017). Domestic sewage rich in sodium
triphosphate (a component in synthetic detergents) can stimulate
the growth and proliferation of Chlorella vulgaris (Marchello
et al., 2015). Cyclotella meneghinianais an
indicator of organic pollution (Krupa et al., 2016).
Euglena sp. is a biological indicator of high organic
pollution (Mahapatra et al., 2011). A shift in
phytoplankton composition towards bloom-forming cyanobacteria
occurs due to excess phosphorus loading (Lv et al., 2011).
Cyanobacteria were found to be dominant in lakes of
Vrishabhavathi valley, Bangalore. Microcystis sp.
belonging to Cyanobacteria formed surface blooms in Doraikere,
Uttarahalli, ISRO layout, and Yediyur lakes. Microcystis can
tolerate high temperatures and hence survive well in warm,
shallow, and eutrophic environments (Ke et al., 2008).
They form blooms in water bodies due to the buoyant nature
imparted by the gas vacuoles (Muthukumar et al., 2007).
The surface blooms of Microcystis sp. will decrease the
water transparency as well as light irradiation to bottom
layers, which in turn inhibits the growth of benthic algae (Su
et al., 2014; Teta et al., 2017). Harmful
algal blooms (HABs) forms surface scum, induce hypoxia, alter
the food web, and produce toxins thus, threatens ecosystem and
human health (Fang et al., 2019; Lu et al., 2013;
Catherine et al., 2013). Excessive phytoplankton growth
eventually causes depletion of dissolved oxygen (DO) and kills
fish as well as other aquatic life. Cyanobacteria produce toxic
secondary metabolites called cyanotoxins, such as hepatotoxins,
dermatotoxins, neurotoxins, and cytotoxins (Du et al., 2019;
Beaver et al., 2018). These cyanotoxins differ in their
chemical structure and toxicology (Merel et al., 2013).
The hepatotoxic microcystins (MCs) produced by
Microcystis sp. blooms at higher water temperature, pH,
and dissolved oxygen (Wu et al., 2014). Generally,
species under Cyanophyceae are resistant to inorganic pollution,
organic pollution, untreated effluents, and anaerobiosis
(Marchello et al., 2015). In Dongping lake, temperature
and chemical oxygen demand (COD) are the main drivers of the
cyanobacterial community composition (Lu et al., 2013).
Cyanobacteria biomass increased with a rise in total phosphorus
concentration (Yang et al., 2016). Microcystis
aeruginosa, Spirulina sp., and Oscillatoria sp.
are bioindicators of eutrophic conditions (Wijeyaratne and
Nanayakkara, 2020). The cyanobacterial dominance was influenced
by rainfall, flow, water temperature, EC, DO, pH, total
nitrogen, nitrate, ammonia, total phosphorus, ortho-phosphate,
chlorophyll–a, BOD, COD, total organic carbon, iron, and silicon
dioxide content (Kim et al., 2019). Cyanophyta grows
faster under conditions such as increased temperature, salinity,
and light intensity with less water turbulence (Kouhanestani
et al., 2019).
Water quality monitoring and management of lakes at regular time
intervals are compulsory to control water pollution and
deterioration of freshwater ecosystems. The integration of
wastewater treatment plants with constructed wetlands and algal
pond helps in efficient nutrient removal from urban lakes
(Ramachandra et al., 2018). The periodic monitoring of
lakes will also aid in bloom management. It is necessary to
control the levels of nutrients, dissolved oxygen, and organic
matter content of water bodies to prevent algal blooms (Chen
et al., 2018). Aeration of water bodies will increase
the dissolved oxygen levels and reduce bloom formation tendency
(Ramachandra et al., 2015). A ban on the use of
phosphate-containing detergents and improved wastewater
treatment can reduce the phosphorus (P) loads in lakes.
Reductions in nutrient (N and P) input are requisite for
effective long- term control of cyanobacterial algal blooms
(Paerl et al., 2011). The increasing water pollution
and harmful cyanobacterial algal blooms due to nutrient
enrichment in lakes pose a severe threat to freshwater resources
and biodiversity.
|