l |
l |
l |
Energy and Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, India
Email: tvr@iisc.ac.in, emram.ces@courses.iisc.ac.in
l |
l |
l |
l |
l |
l |
Results and discussion
3.1 Hydrological Variations of Lentic and Lotic Systems
Estuaries are dynamic ecosystems with diurnal and seasonal variations in hydrological and environmental parameters. However, in-situ parameters of temperature and salinity did not reveal a drastic variation as the sampling was done for a season – pre-monsoon in both lentic and lotic systems. The average air temperature across different habitats was 30.85 ± 2.60oC. Water temperature is an important parameter that influences many abiotic chemical processes like dissolution – precipitation, oxidation – reduction, adsorption – desorption and physiology of biotic community in a habitat (van Aken 2008). It also influences the rate of photosynthesis of an aquatic system (Fatema et al. 2014, Yin 2002). All the eight lentic and lotic habitats had warm waters and high salinity, as sampling was done during summer months of the year. The changes in water temperature are influenced by solar insolation, fresh water influx, evaporation, cooling and mix up with ebb and flow from adjoining neritic waters (Madhu et al. 2007). The average water temperature recorded across the habitats was 31.21 ± 2.61oC. In comparison 31 - 32oC during March-April at the Tapi estuarine region, Gulf of Khambhat, Indian west coast (Cochin estuary) by George et al. 2012, Martin et al. 2011 and Parangipettai (south-east coast of India) by Godhantaraman 2002 reported similar surface water temperature during pre-monsoon season with a variation range of 1 – 2oC. The pH is a master variable that determines many chemical, biological and kinetic processes in natural waters (Millero 1986). The pH of unpolluted natural waterbodies can broadly vary between 3 – 11, however, pH of those water bodies are in the range of 5 – 9, which generally supports diverse assemblage of aquatic species (Alabaster et al. 1984) . The pH remained alkaline at all the habitats 7.9 ± 0.09 except for salt marsh sedges (SMS) study station which showed an acidic pH due to hypoxic environment with very low DO level due to dead and decaying organic matter. Satpathy et al. 2010 observed a pH range of 7.7 to 8.3 along the Kalpakkam estuarine waters of southeast coast of India. Madhu et al. 2007 observed a steady pre-monsoon pH of 7.1 ± 0.14 in Kochi backwaters corroborating well with the present study.
Salinity defines the relative proportions of fresh and saline waters found in different parts of the estuary. Salinity levels of a region are influenced by various factors such as the location of sampling station in the estuary, the daily tides and the volume of fresh water flowing into the estuary. Generally during pre-monsoon, salinity rises due to higher temperatures and increased evaporation rate (Mann 2000, Sumich & Morrissey 2004, Levinton 2017)As the sampling was done during the pre-monsoon season, salinity of both lentic and lotic habitats exhibited similar salinity levels (27.66± 1.50 ppt), except saltpan which had hypersaline condition of 58 ppt. Madhu et al. 2007 found salinity of 30 ppt during the pre-monsoon season in Cochin estuary, which matches well with the salinity ranges of the present study.
Dissolved oxygen is a critical parameter that determines the ability of an aquatic system to support aquatic biota. The average DO levels of lotic habitats (MF, KOSB, BRS, ERS) were 7.15± 0.18 mg/L, which are slightly higher when compared to lentic habitats. Among the lentic habitats, salt pan (SP) and salt marsh sedges (SMS) had the least DO levels of 2.70 ± 0.09 mg/L due to extreme salinity at the salt pan and hypoxic condition and very low water level at the salt marsh sedges respectively. Increased salinity is known to reduce the dissolution of oxygen in water thus reducing the DO levels drastically. This could be the reason behind saltpan station exhibiting very low DO levels. The DO values in lotic habitats were higher than the DO values reported in many other Indian estuaries. George et al. 2012 observed a lower level of DO in Tapi estuary, at 5.8 ± 0.9 mg/L, during pre-monsoon season which was attributed to higher levels of domestic and industrial effluents. The DO of Aganashini sites were higher than the DO level recorded in the Tuticorin estuary by Balakrishnan et al. 2017. However, lower values of DO were reported earlier for similar ecosystems in tropical estuaries of India and South-east Asia by Madhu et al. 2007, Martin et al. 2011, Ouyang et al. 2006 and Juahir et al. 2011, which could be attributed to the large influx of domestic and industrial effluents in those estuaries. In comparison, the Aganashini estuary is more pristine with neither industrial establishments in the vicinity, nor any major township. Estuarine paddy field (APF) and Bargi gazni (BG) sites exhibited moderate DO levels than other two lentic habitats where DO averaged 7.08 ± 1.0 mg/L. This Higher level of DO could be due to the inflow of more fresh water, since all the lotic habitat stations were near upstream of the estuary.
Nitrates (NO3--N) the oxidized form of nitrogen, is also an indicator of the level of anthropogenic stress. Nutrients especially nitrates (NO3- -N), and silicates (SiO44-) showed wider fluctuations in their ranges across different habitats whereas the phosphate variations were comparatively marginal across habitats (Table 2). The phosphate values varied within a range of 0.21 mg/L to 2.38 mg/L. Mud flats (MF), Egret roosting site (ERS) and Bat roosting site (BRS) had very high nitrate levels of 8.41±0.30 mg/L, 9.82 ±0.97 mg/L and 6.72±0.20 mg/L respectively due to site-specific litter loadings from mangroves and sedges as well as leaf litter and faecal droppings of faunal species. The mudflats make a prominent intertidal region, constituting the main habitat of Paphia malabarica, the dominant bivalve which is harvested in bulk during pre-monsoon, in addition to six other bivalve species in lesser quantities (Boominathan et al. 2008). These bivalves are filter feeders that feed on phyto and zooplankton which are higher in these regions, more in the pre-monsoon. Additionally, large flocks of migratory and wintering shorebirds like sea gulls and egrets also visit these habitats. Thus, the organic debris loading from the dead bivalves, phytoplankton and bird droppings and the excretes of the bivalves, have contributed to higher nitrates in the mudflats. Bat and egret roosting sites also had higher nitrate levels due to droppings.
The level of silicates is a notable determinant of primary productivity and the main sources of silicates in estuaries are through land-based run-offs during monsoon. Silicate (SiO44-) levels in estuarine and coastal regions are driven by factors such as physical mixing of fresh and saline waters, siltation in the upstream, rock weathering, adsorption from sedimentary particles, and nutrient upwelling that results in chemical interaction with deep clayey sediments and biological fixation by phytoplankton’s, especially by diatoms and silicoflagellates (Richardson et al. 2000, Shah et al. 2008, Prabu et al. 2008, Satpathy et al. 2010). The silicate in all the stations, covering lentic and lotic habitats, ranged from 4.17 mg/L to 10.03 mg/L; mudflats exhibited the highest (10.03 ± 0.48 mg/L) whereas in all lotic habitats the silicates averaged 4.2 ± 0.06 mg/L. Lentic habitats of APF and BG showed silicate values averaging 4.4 ± 0.29 mg/L, like BRS and ERS. Salt pan and salt marsh sedges had a silicate concentration of 7.34± 0.20 mg/L and 5.92 ±0.13 mg/L respectively. When the overall seasonality of silicate levels in other estuaries are considered, Martin et al. 2008, Satpathy et al. 2010, Balakrishnan et al. 2017 reported comparatively lesser silicate values during the pre-monsoon. These nutrient values of lentic and lotic systems in the Aganashini estuary vary, as it is relatively an isolated system with wide variations in habitat conditions and trophic status having higher nutrient loadings. The physico-chemical parameters recorded at different stations are detailed in Table 2.
| Sites | AT(°C) | WT(°C) | pH | DO(mg/L) | Salinity(ppt) | N-NO3-(mg/L) | P-PO43-(mg/L) | SiO4-4(mg/L) |
| SP(Ln) | 32 | 31 | 8 | 2.64±0.03 | 58 | 0.56±0.03 | 0.21±0.02 | 7.34±0.20 |
| SMS (Ln) | 25 | 25.5 | 6.8 | 2.77 ± 0.64 | 27 | 2.86 ± 0.22 | 1.4 ± 0.34 | 5.92 ± 0.13 |
| BG (Ln) | 34 | 33 | 7.9 | 6.37 ± 0.31 | 23 | 2.38 ± 0.27 | 0.91 ± 0.45 | 4.19 ± 0.10 |
| MF (Lt) | 32 | 32 | 7.9 | 7.11 ± 0.14 | 29 | 8.41 ± 0.30 | 1.31 ± 0.26 | 10.03 ± 0.48 |
| KOSB (Lt) | 31 | 32 | 8.0 | 7.02 ± 0.08 | 25 | 2.11 ± 0.04 | 0.86 ± 0.04 | 4.17 ± 0.15 |
| BRS (Lt) | 32 | 33 | 7.8 | 7.43 ± 0.51 | 28 | 6.72 ± 0.20 | 0.53 ± 0.15 | 4.24 ± 0.10 |
| ERS (Lt) | 32 | 33 | 7.8 | 7.06 ± 0.07 | 28 | 9.82 ± 0.97 | 0.33 ± 0.07 | 4.46 ± 0.42 |
| APF (Ln) | 32 | 32 | 7.8 | 7.79 ± 0.17 | 29 | 3.12 ± 0.12 | 0.53 ± 0.09 | 4.61 ± 0.27 |
3.2 Understanding the Species Dynamics – a prelude to phyco-prospecting
Prioritizing the diatom strains based on their response to fluctuating environmental conditions would entail understanding species at local levels. Choosing a diatom strain or a consortium of strains that are consistently productive across seasons under fluctuating environmental conditions would be more advantageous in realizing sustainable biofuel production using microalgae (Hildebrand et al. 2012). Hence, understanding the species distribution and its tolerance levels would give insights for choosing a diatom strain as a potential biofuel feedstock. A total of 80 different species were recorded from all the habitats with species richness varying from 6 to 29 across sampling sites. The spatial distribution of diatom species of lentic and lotic habitats is depicted in Fig. 2 (a-b). The similarity of diatom assemblages among the replicates collected from the same stations were high. Salt pan (SP) had the lowest diversity with only six species, while relative abundance was the highest with 16.67% followed by Oyster shell bed (KOSB) site where species richness and relative abundance were 7 and 14.28% respectively. Bargi gazni had a species richness of 12 and a relative abundance of 8.33%.
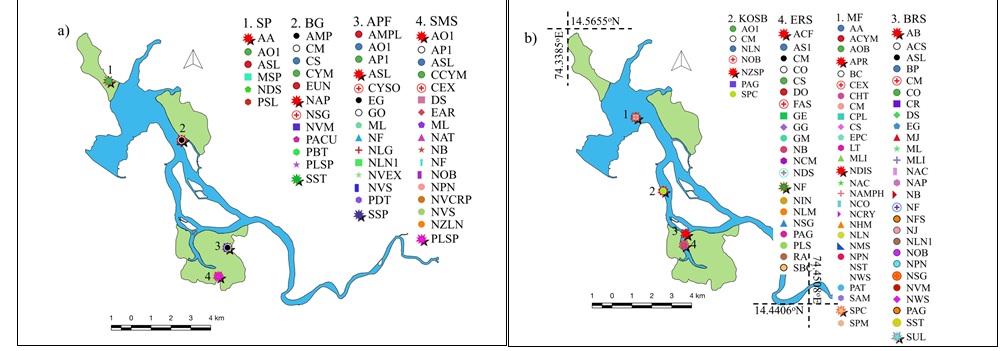
Fig. 2 (a – b) Spatial Mapping of Diatoms in Lentic and Lotic Habitats
A total of 27 out of 80 species were present at least in minimum two to maximum five different habitats whereas the remaining 53 species were highly endemic as well as unique to only one of the eight habitats. The 27 most prevalent species could be considered as more of a cosmopolitan kind of species that have adapted with higher resilience in widely varying nutrient and habitat conditions. Amphora salina, Amphora ovalis, Cyclotella meneghiniana, Navicula forcipata, Nitzschia obtusa and Pleurosigma angulatum exhibited cosmopolitan nature with their presence in at least four out of total eight habitats. Species like Bacteriastum cosmosum, Achnanthes oblongella, Amphora cymbifera, Cocconeis pelta, Navicula weissflogii, N. amphisbaena, N. scutelloids, Eunotia pectinalis, Licomophora tincta and Sellaphora americana were confined to only mudflats, with their relative abundances of <5%. These species could be considered as sensitive to habitat changes, their presence being governed by one or more of the prevailing favorable environmental conditions. Fig. 3 reflects composition of species distributed across different habitats.
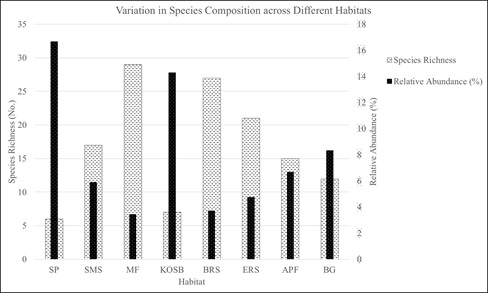
Fig. 3 Graph Showing Species Richness and Relative Abundance across Habitats
3.3 Site-wise Variation in Species Dynamics
Habitat condition plays a vital role in determining the presence or absence of a species or a group of species in a habitat depending on the level of dominance and tolerance to the prevailing environmental conditions. Diversity and dominance are determined using Shannon – wiener’s Diversity (H’) and Simpson’s dominance (D) indices using the species richness data of the epipelic diatoms collected from sampling locations. Higher diatom diversity was found in mudflats (MF) (H’ = 3.25) followed by bat roosting site (BR) (H’ = 3.13). Least diversity was observed in the salt pan (SP) station (H’ = 1.7) followed by Kagal Oyster shell beds (KOSB) (H’ = 1.82). Salt marsh sedges (SMS), egret roosting site (ERS), abandoned paddy fields (APF) and Bargi gazni (BG) had almost equal diversity ranges between 2.42 to 2.91. Simpson’s dominance index yielded higher values for SP and KOSB station indicating the existence of highly tolerant dominant species. MF and BRS stations had the least dominance index (D = 0.04 and D = 0.05) and rich species diversity (Fig. 4), confirming inverse relationship of diversity and dominance at the respective sampling locations.
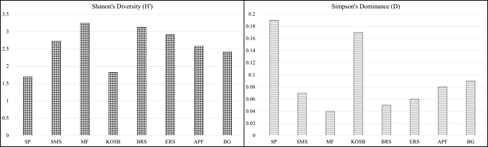
Fig. 4 Diversity and Dominance indices of diatoms from different habitats
3.4 Influence of Nutrients on Species Presence
The level of significance of nutrient composition in determining the species presence and abundance at a region is evaluated using ANOVA, which indicates F = 6.864 (p<0.05) and df = 11.83 of nitrate and silicate levels on species richness. Turkey’s pairwise comparisons on nitrate and silicate levels to species richness showed a higher level of significance (p <0.01) (Krzywinski & Altman 2013) with stress level of 1% with p = 0.0006 for nitrates and p= 0.001 for silicates. This highlights that diatom species composition depends on the levels of nitrates and silicates at the habitat.
3.5 Understanding the influence of habitat conditions on species distribution
Diatom growth at a location depends on salinity, temperature and pH in addition to nutrients. The similarities among the habitats with respect to nutrient fluxes and species composition is assessed through a multivariate hierarchical clustering analysis. Clustering of study regions was done using unpaired group mean average algorithm (UPGMA) using PAST V 3.0. The study stations were grouped / clustered based on their relative similarity between the environmental conditions and species commonness among the regions. Saltpan having extreme salinity and poor nutrient conditions had shown the least similarity with other stations which is evident from the highest distance index in the generated dendrogram, followed by salt marsh sedges which exhibited hypoxic conditions due to dead and decaying organic matter. The shortness of the length of the arms between Bargi gazni and Kagal oyster shell bed in the dendrogram highlights the commonness in the environmental parameters and species richness. Hierarchical cluster dendrogram (Fig. 5) illustrates a higher similarity in nutrient ranges and species compositions among bat roosting site and abandoned paddy fields with egret roosting site and mudflats habitats.
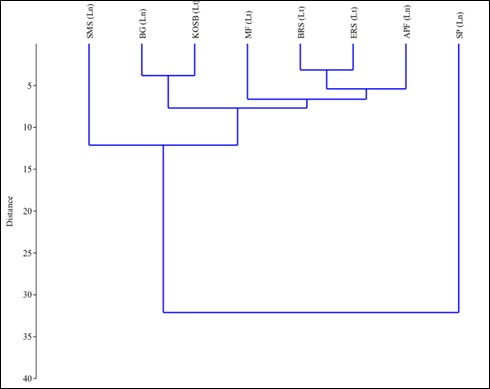
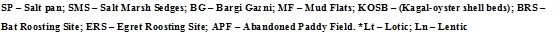
Fig. 5 Multivariate Hierarchical Clustering of different habitats
3.6 Quest for Tolerant Species
Understanding the interactions of habitat and environmental parameters on species composition gains importance in determining the tolerance level of a species for a sustainable future biofuel production system. Tolerance is developed when a species is exposed to prolonged unfavorable conditions, the species tends to build up resilience to withstand harsh environmental conditions. The combination of environmental variables that has significant influence on the dispersion of the scores of species (Nabout et.al 2006) is chosen through CCA - ordination technique. CCA ordination was done considering the biological parameters obtained from all the eight stations having diverse habitat conditions with the varied environmental parameters to understand the level of influence of each of the environmental parameters and habitat conditions on diatom species composition and the results are presented in Fig. 6. Axis 1 represents the environmental variables of phosphates and silicates, while air temperature, water temperature and pH gradients are represented by axis 2 (Table 3). The stations Mudflats (MF) and Kagal-oyster shell beds (KOSB) were highly oriented towards axis 1, which demonstrated the influence of phosphate and silicate levels in their corresponding species composition variations in these two lotic habitat stations. Navicula lanceolata, Stauroneis pachycephala, Nitzschia acicularis, Surirella striatula and Melosira lineatus were strongly influenced by the levels of phosphates and silicates in MF and KOSB stations.
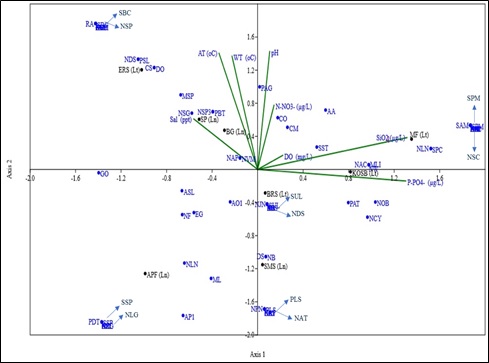
Fig. 6 CCA triplot showing relationship between environmental variables and diatom species composition (Acronyms of the species names given in Annexure 1)
| Estimate | SE | t value | p | |
| Intercept | - 5372.14 | 1562.89 | 3.34 | 0.001 |
| X1 | - 1.86 | 0.86 | - 2.17 | 0.036 |
| X2 | 15.42 | 7.33 | 2.10 | 0.042 |
| X3 | - 5.47 | 2.09 | - 2.61 | 0.013 |
| X4 | 7.09 | 2.42 | 2.91 | 0.006 |
| X5 | - 91.73 | 40.40 | - 2.27 | 0.029 |
| X6 | 250.31 | 82.98 | 3.01 | 0.004 |
| X7 | 103.35 | 44.87 | 2.30 | 0.027 |
Table 3. Axis Scores of Physico-Chemical Variables in CCA Ordination (P<0.05)
The lentic habitats - salt pan (SP) and Bargi gazni (BG) depicted a strong positive correlation with salinity, air and water temperature. The presence of Pleurosigma balticum, Melosira species, Nitzschia sigma and Nitzschia spp. in SP and BG stations were highly influenced by salinity, air and water temperature. The presence of Pleurosigma angulatum in different stations (KOSB, BRS, ERS and BG) was governed by the changes in pH, air and water temperature and moderately influenced by nitrates, DO and salinity. Bat roosting site (a lotic habitat) had a strong negative correlation with air temperature, water temperature and salinity. The diatom species Navicula johnsonii, Synedra ulna, Nitzschia fasciola, Diploneis smithi and Nitzschia obtusa exhibited a strong negative correlation with air and water temperature, pH and salinity. The species composition of Cyclotella operculata, Cyclotella meneghiniana and Amphiprora alata were strongly driven by the variation in nitrate levels at different study stations. The cosmopolitan species Amphora salina, Amphora ovalis, Epithema gibberula and Navicula forcipata are present in more than two sampling stations irrespective of the variations in the nutrients and in-situ physico-chemical parameters. Pleurosigma delicatulum, Stauroneis sp., Navicula longicephala and Amphiprora paludosa were locally endemic/unique being restricted to abandoned paddy fields (APF) and Raphoneis amphiceros, Nitzschia longissima and Sellaphora bacilloides were unique to egret roosting site (ERS). The light micrographs of processed diatom frustules after acid digestion are given in Fig. 7.

Fig. 7 Light Micrographs of Processed Diatom Frustules A) Diploneis ovalis B) Navicula expansa C) Navicula weissflogii D) Surirella tenera E) Nitzschia obtusa F) Gomphonema sp. G) Navicula forcipata H) Achnanthes sp. I) Amphiprora sp. J) Stauroneis sp. K) Cymbella sp. L) Cyclotella meneghiniana M) Navicula sp. N) Stauroneis sp. O) Bacillaria paradoxa P) Cymbella sp. Q) Epithema gibberula R) Licomophora sp. S) Gyrosigma eximum T) Amphora sp. U) Navicula cryptocephala V) Gyrosigma Marcum W) Pleurosigma angulatum X) Amphora sp. Y) Navicula pusila
The diatom species compositions across different habitats with varied nutrient levels reveal preference of tolerant and sensitive species to a particular habitat. The species presence, relative abundances and CCA ordination of species with environmental parameters would aid in estimating the relative abundance and sensitiveness at a given study site. For instance, Amphora salina, Amphora ovalis, Epithema gibberula, Cyclotella meneghiniana, Coscinodiscus subtilis, Nitzschia obtusa, Pleurosigma angulatum, Navicula forcipata, Nitzschia panduriformis and Nitzschia sigma were present at least in four of the eight different habitat stations with >10% of relative abundances. This highlights the tolerance and versatility of species endowed with resilience to survive in fluctuating and dynamic environmental conditions. Earlier studies on Amphora sp., Nitzschia sp. had reflected heterotrophic abilities. Nitzschia sp. was found to be obligatory heterotrophic in habitats strongly favoring heterotrophic growth on decaying piles of seaweeds with the lower light penetration and higher organic substrate (Linkins 1973). The centric Cyclotella meneghiniana was also known to possess heterotrophic capabilities and was found to be the most dominant in a sewage maturation pond (Schoeman 1972, Schoeman 1979). Pleurosigma angulatum and Coscinodiscus subtilis, typical marine species, were abundant in more than 4 habitats, with higher relative abundances of 15.4% and 9.4% respectively. Desrosieres 1969 reported Coscinodiscus sp. as a strong eutrophic indicator. Round 1991 had reported Pleurosigma sp. and Amphora sp. as epipelic diatoms found predominantly on mudflats. Targeting such obligatory heterotrophic and naturally abundant species under rugged environmental conditions for biofuel production would greatly reduce the risk of contamination and yield higher productivity.
On the contrary, Sellaphora Americana, Sellaphora bacilloides, Nitzschia longissima, Raphoneis amphiceros, Navicula longicephala, Spermatogonia sp., and Stauroneis sp., Pleurosigma salinarum and Nitzschia dissipata were found at only one location with the lower relative abundance (<5%). As these species occurred sparsely and showing insignificant correlation with varying physico-chemical parameters, these diatom taxa are sensitive, surviving in only locations with favorable environmental conditions. Insights of the variations in diatom community structure in terms of species abundance, tolerance and sensitivity would aid in selecting a candidate strain or a consortium available indigenously for scale up towards sustainable biofuel production.
3.7 Multivariate Analyses of Habitat Conditions for Estimating Lipid Productivity Potential
Lipid productivity and habitat condition details were also compiled from published literatures (Barclay et al. 2007, Sheehan et al. 1998, Potapova & Charles 2002, Weilhoefer & Pan 2006, De La Peña 2007, Taylor, Prygiel, et al. 2007, Besse-Lototskaya et al. 2011, Chen 2012, Delgado et al. 2012, Fore & Grafe 2002, Rimet 2009, Scholz & Liebezeit 2013, Tan et al. 2014, D’Ippolito et al. 2004, d’Ippolito et al. 2015, Zhao et al. 2016, Hausmann et al. 2016, Fields & Kociolek 2015, Tan et al. 2017). Agglomerative hierarchical clustering (Fig. 8) was carried out relating environmental conditions to the lipid content, which indicated four distinct diatom species clusters. Species such as Achnanthes sp., Melosira sp., Navicula sp., and Nitzschia sp., forming one dominant cluster (cluster:4) seems to be the potential lipid accumulators. Next, dominant cluster (cluster:2) consists of sensitive species like Synedra sp., Cocconeis sp., Diploneis sp., Gyrosigma sp., some sensitive species of Nitzschia sp., and Navicula sp. which are similar to the current study. Cluster 3 grouped the planktonic marine diatoms of Chaetoceros sp., Thalassiosira sp., Skeletonema sp., and Phaeodactylum tricornutum indicates a clear distinction in its lipid content and other growth characteristics when compared to benthic diatom forms. Cluster 1 is formed by only two species Amphiprora hyalina, Nitzschia dissipata that were characterized by its projected lipid productivity at higher silicate concentrations and less light intensity. Thus, the cluster analysis provided insights towards phyco-prospecting of algal species based on its preference to environmental parameters as well as their lipid content. Multivariate regression of environmental parameters with lipid content gave probable relationship with the significant coefficient of determination (R) at p<0.05 for nutrients - Nitrates (p=0.036), Phosphates (p=0.042), Silicates (p=0.013) as well as Salinity (p=0.029) and pH (p=0.027). Correlation with p<0.01 was observed for temperature (p=0.004) and light intensity (p= 0.006). The overall p of the linear regression was found to be p=0.043. The estimated coefficients, standard error, t and p values of each of the independent variables are given in Table 4.
Y= -1.868(X1 )+15.423(X2 )-5.478(X3 )+7.091(X4 )-91.734(X5 )+250.31(X6 )+103.35(X7 )-5372.14 …1
Where, Y = Lipid content in %dcw; X1= Nitrate concentration in mg/L; X2= Phosphate concentration in mg/L; X3= Silicate concentration in mg/L; X4= Light intensity (µmol m-2 S-1); X5= Salinity (ppt); X6= Temperature (°C); X7= pH
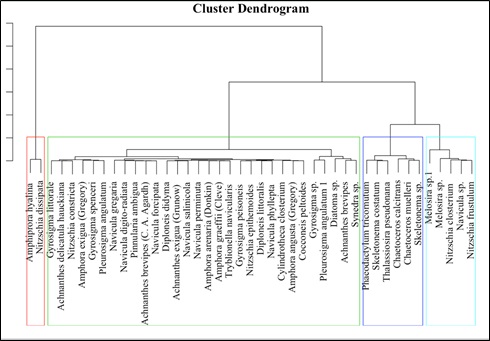
Fig. 8 Agglomerative Hierarchical Clustering based on Critical Growth Parameters and Lipid Content
| Variables | Axis 1 (k = 0.719) | Axis 2 (k = 0.682) |
| Air temperature | - 0.135 | 0.561 |
| Water Temperature | - 0.090 | 0.547 |
| pH | 0.042 | 0.570 |
| Salinity | - 0.224 | 0.243 |
| DO | 0.088 | 0.068 |
| Nitrates | 0.057 | 0.310 |
| Phosphates | 0.520 | - 0.055 |
| Silicates | 0.524 | 0.15 |
Table 4. Coefficients, std. Error, t and p value of Multivariate Regression Analysis
The equation 1 helps in determining the lipid content of select diatoms considering the environmental parameters of a location. It is evident from the literatures that only very few species belonging to common diatom genus like Amphora, Amphiprora, Cocconeis, Chaetoceros, Diploneis, Melosira, Pleurosigma, Navicula, Nitzschia, Gyrosigma, Thalassiosira and Skeletonema have been investigated under different stress conditions while many species are still unexplored. This empirical model (equation 1) aids in estimating the lipid indirectly based on the site conditions as well as species type and could be used as screening criteria for candidate strain selection for large scale biofuel production. The current research unravels the unexplored potential diatoms, especially salt tolerant marine diatoms which gives scope for further research on these indigenous species for extraction of both lipids as well as value added products. Thus, the study could be considered as an essential prelude to phyco-prospecting indigenous diatom strains that are capable of accumulating higher lipids even at fluctuating environmental conditions. The statistical analyses revealed a set of tolerant species that can withstand a wide range of nutrient fluctuations, a characteristic feature beneficial for large-scale outdoor cultivation. If such tolerant species are targeted for scale-up studies, either as single strain or a consortium of tolerant strains would certainly avert the risks of contamination due to open air exposure. Moreover, the empirical equation (equation 1) generated would aid in the assessment of lipid content that can be used for screening a potential candidate diatom strain without performing expensive lab-scale cultivation using synthetic media.
| * Corresponding Author : | |||
| Dr. T.V. Ramachandra Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. Tel : 91-80-23600985 / 22932506 / 22933099, Fax : 91-80-23601428 / 23600085 / 23600683 [CES-TVR] E-mail : tvr@iisc.ac.in, emram.ces@courses.iisc.ac.in, energy@ces.iisc.ac.in, Web : http://wgbis.ces.iisc.ac.in/energy |
|||
| * Corresponding Author : | |||
| Dr. T.V. Ramachandra Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560 012, INDIA. Tel : 91-80-23600985 / 22932506 / 22933099, Fax : 91-80-23601428 / 23600085 / 23600683 [CES-TVR] E-mail : cestvr@ces.iisc.ac.in, energy@ces.iisc.ac.in, Web : http://wgbis.ces.iisc.ac.in/energy |
|||
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560012.
Centre for Sustainable Technologies, Indian Institute of Science, Bangalore – 560012.
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560012.
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560012.
Energy & Wetlands Research Group, Centre for Ecological Sciences, Indian Institute of Science, Bangalore – 560012.
Centre for Sustainable Technologies, Indian Institute of Science, Bangalore – 560012.


